Pneumococcal Vaccine 13-Valent
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Alonso Alvarado, M.D. [2]
Disclaimer
WikiDoc MAKES NO GUARANTEE OF VALIDITY. WikiDoc is not a professional health care provider, nor is it a suitable replacement for a licensed healthcare provider. WikiDoc is intended to be an educational tool, not a tool for any form of healthcare delivery. The educational content on WikiDoc drug pages is based upon the FDA package insert, National Library of Medicine content and practice guidelines / consensus statements. WikiDoc does not promote the administration of any medication or device that is not consistent with its labeling. Please read our full disclaimer here.
Overview
Pneumococcal Vaccine 13-Valent is a vaccine that is FDA approved for the prophylaxis of infecetions caused by S. pneumoniae serotypes 1, 3, 4, 5, 6A, 6B, 7F, 9V, 14, 18C, 19A, 19F and 23F in children 6 weeks to 17 years-old and adults 50 years old and older. Common adverse reactions include erythema at injection site, injection site pain, swelling at injection site, tenderness, decrease in appetite, aggravated and new onset arthralgia, difficulty moving arm, aggravated and new onset myalgia, decreased sleep, headache, hypersomnia, irritability, fatigue..
Adult Indications and Dosage
FDA-Labeled Indications and Dosage (Adult)
Preparation for Administration
Since this product is a suspension containing an adjuvant, shake vigorously immediately prior to use to obtain a homogenous, white suspension in the vaccine container. Do not use the vaccine, if it cannot be resuspended. Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration. This product should not be used if particulate matter or discoloration is found.
Do not mix Prevnar 13 with other vaccines/products in the same syringe.
Administration Information
For intramuscular injection only.
Each 0.5 mL dose is to be injected intramuscularly using a sterile needle attached to the supplied prefilled syringe. The preferred sites for injection are the anterolateral aspect of the thigh in infants and the deltoid muscle of the upper arm in toddlers, children and adults. The vaccine should not be injected in the gluteal area or areas where there may be a major nerve trunk and/or blood vessel.
Vaccination Schedule for Adults 50 years of Age and Older
Prevnar 13 is administered as a single dose.
Off-Label Use and Dosage (Adult)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Pneumococcal Vaccine 13-Valent in adult patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Pneumococcal Vaccine 13-Valent in adult patients.
Pediatric Indications and Dosage
FDA-Labeled Indications and Dosage (Pediatric)
Preparation for Administration
Since this product is a suspension containing an adjuvant, shake vigorously immediately prior to use to obtain a homogenous, white suspension in the vaccine container. Do not use the vaccine, if it cannot be resuspended. Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration [see Description (11)]. This product should not be used if particulate matter or discoloration is found.
Do not mix Prevnar 13 with other vaccines/products in the same syringe.
Administration Information
For intramuscular injection only.
Each 0.5 mL dose is to be injected intramuscularly using a sterile needle attached to the supplied prefilled syringe. The preferred sites for injection are the anterolateral aspect of the thigh in infants and the deltoid muscle of the upper arm in toddlers, children and adults. The vaccine should not be injected in the gluteal area or areas where there may be a major nerve trunk and/or blood vessel.
Vaccination Schedule for Infants and Toddlers
Prevnar 13 is to be administered as a four-dose series at 2, 4, 6, and 12–15 months of age.

Vaccination Schedule for Unvaccinated Children 7 Months Through 5 Years of Age
For children 7 months through 5 years of age who have not received Prevnar® or Prevnar 13, the catch-up schedule in Table 2 applies:

The immune responses induced by this catch-up schedule may result in lower antibody concentrations for some serotypes, compared to antibody concentrations following 4 doses of Prevnar 13 (given at 2, 4, 6, and 12–15 months). In children 24 months through 5 years of age, lower antibody concentrations were observed for some serotypes, compared to antibody concentrations following 3 doses of Prevnar 13 (given at 2, 4, and 6 months).
Vaccination Schedule for Children Previously Vaccinated With Prevnar Pneumococcal 7-valent Conjugate Vaccine (Diphtheria CRM197 Protein)
Children 15 months through 5 years of age who are considered completely immunized with Prevnar may receive one dose of Prevnar 13 to elicit immune responses to the six additional serotypes. This catch-up (supplemental) dose of Prevnar 13 should be administered with an interval of at least 8 weeks after the final dose of Prevnar. The immune responses induced by this Prevnar 13 schedule may result in lower antibody concentrations for the 6 additional serotypes (types 1, 3, 5, 6A, 7F, and 19A), compared to antibody concentrations following 4 doses of Prevnar 13 (given at 2, 4, 6, and 12–15 months).
Vaccination Schedule for Children 6 Years Through 17 Years of Age
In children 6 years through 17 years of age, Prevnar 13 is administered as single dose. If Prevnar was previously administered, then at least 8 weeks should elapse before receiving Prevnar 13.
Off-Label Use and Dosage (Pediatric)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Pneumococcal Vaccine 13-Valent in pediatric patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Pneumococcal Vaccine 13-Valent in pediatric patients.
Contraindications
Severe allergic reaction (e.g., anaphylaxis) to any component of Prevnar 13 or any diphtheria toxoid-containing vaccine.
Warnings
Management of Allergic Reactions
Epinephrine and other appropriate agents used to manage immediate allergic reactions must be immediately available should an acute anaphylactic reaction occur following administration of Prevnar 13.
Altered Immunocompetence
Individuals with altered immunocompetence, including those at higher risk for invasive pneumococcal disease (e.g., individuals with congenital or acquired splenic dysfunction, HIV infection, malignancy, hematopoietic stem cell transplant, nephrotic syndrome), may have reduced antibody responses to immunization with Prevnar 13.
Apnea in Premature Infants
Apnea following intramuscular vaccination has been observed in some infants born prematurely. Decisions about when to administer an intramuscular vaccine, including Prevnar 13, to infants born prematurely should be based on consideration of the individual infant's medical status and the potential benefits and possible risks of vaccination.
Adverse Reactions
Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse-reaction rates observed in the clinical trials of a vaccine cannot be directly compared to rates in the clinical trials of another vaccine and may not reflect the rates observed in practice. As with any vaccine, there is the possibility that broad use of Prevnar 13 could reveal adverse reactions not observed in clinical trials.
Clinical Trials Experience With Prevnar 13 in Children 6 Weeks Through 17 Years of Age
The safety of Prevnar 13 was evaluated in 13 clinical trials in which 4,729 infants (6 weeks through 11 months of age) and toddlers (12 months through 15 months of age) received at least one dose of Prevnar 13 and 2,760 infants and toddlers received at least one dose of Prevnar active control. Safety data for the first three doses are available for all 13 infant studies; dose 4 data are available for 10 studies; and data for the 6-month follow-up are available for 7 studies. The vaccination schedule and concomitant vaccinations used in these infant trials were consistent with country-specific recommendations and local clinical practice. There were no substantive differences in demographic characteristics between the vaccine groups. By race, 84.0% of subjects were White, 6.0% were Black or African-American, 5.8% were Asian and 3.8% were of 'Other' race (most of these being biracial). Overall, 52.3% of subjects were male infants.
Three studies in the US (Studies 1, 2 and 3) evaluated the safety of Prevnar 13 when administered concomitantly with routine US pediatric vaccinations at 2, 4, 6, and 12–15 months of age. Solicited local and systemic adverse events were recorded daily by parents/guardians using an electronic diary for 7 consecutive days following each vaccination. For unsolicited adverse events, study subjects were monitored from administration of the first dose until one month after the infant series, and for one month after the administration of the toddler dose. Information regarding unsolicited and serious adverse events, newly diagnosed chronic medical conditions, and hospitalizations since the last visit were collected during the clinic visit for the fourth-study dose and during a scripted telephone interview 6 months after the fourth-study dose. Serious adverse events were also collected throughout the study period. Overall, the safety data show a similar proportion of Prevnar 13 and Prevnar subjects reporting serious adverse events. Among US study subjects, a similar proportion of Prevnar 13 and Prevnar recipients reported solicited local and systemic adverse reactions as well as unsolicited adverse events.
Serious Adverse Events in All Infant and Toddler Clinical Studies
Serious adverse events were collected throughout the study period for all 13 clinical trials. This reporting period is longer than the 30-day post-vaccination period used in some vaccine trials. The longer reporting period may have resulted in serious adverse events being reported in a higher percentage of subjects than for other vaccines. Serious adverse events reported following vaccination in infants and toddlers occurred in 8.2% among Prevnar 13 recipients and 7.2% among Prevnar recipients. Serious adverse events observed during different study periods for Prevnar 13 and Prevnar respectively were: 1) 3.7% and 3.5% from dose 1 to the bleed approximately 1 month after the infant series; 2) 3.6% and 2.7% from the bleed after the infant series to the toddler dose; 3) 0.9% and 0.8% from the toddler dose to the bleed approximately 1 month after the toddler dose and 4) 2.5% and 2.8% during the 6 month follow up period after the last dose.
The most commonly reported serious adverse events were in the 'Infections and infestations' system organ class including bronchiolitis (0.9%, 1.1%), gastroenteritis, (0.9%, 0.9%), and pneumonia (0.9%, 0.5%) for Prevnar 13 and Prevnar respectively.
There were 3 (0.063%) deaths among Prevnar 13 recipients, and 1 (0.036%) death in Prevnar recipients, all as a result of sudden infant death syndrome (SIDS). These SIDS rates are consistent with published age specific background rates of SIDS from the year 2000.
Among 6,839 subjects who received at least 1 dose of Prevnar 13 in clinical trials conducted globally, there was 1 hypotonic-hyporesponsive episode adverse reaction reported (0.015%). Among 4,204 subjects who received at least 1 dose of Prevnar in clinical trials conducted globally, there were 3 hypotonic-hyporesponsive episode adverse reactions reported (0.071%). All 4 events occurred in a single clinical trial in Brazil in which subjects received whole cell pertussis vaccine at the same time as Prevnar 13 or Prevnar.
Solicited Adverse Reactions in the Three US Infant and Toddler Studies
A total of 1,907 subjects received at least 1 dose of Prevnar 13 and 701 subjects received at least 1 dose of Prevnar in the three US studies (Studies 1, 2 and 3). Most subjects were White (77.3%), 14.2% were Black or African-American, and 1.7% were Asian; 79.1% of subjects were non-Hispanic and non-Latino and 14.6% were Hispanic or Latino. Overall, 53.6% of subjects were male infants.
The incidence and severity of solicited adverse reactions that occurred within 7 days following each dose of Prevnar 13 or Prevnar administered to US infants and toddlers are shown in Tables 3 and 4.
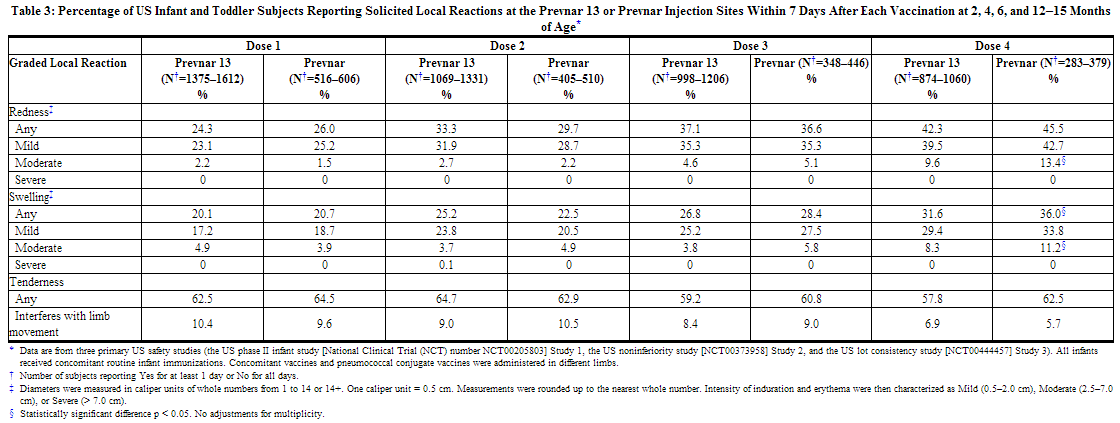
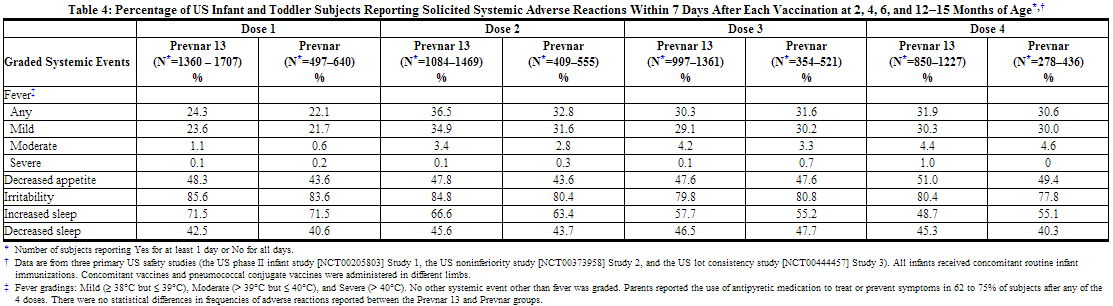
The incidence rates of any fever (≥ 38.0°C) were similar on days 1 and 2 following each dose of Prevnar 13 compared to after each dose of Prevnar administered to US infants and toddlers (day 1 = day of vaccination). After dose 1, fever was reported in 11.0–12.7% on day 1 and 6.4–6.8% on day 2. After dose 2, fever was reported in 12.3–13.1% on day 1 and 12.5–12.8% on day 2. After dose 3, fever was reported in 8.0–9.6% on day 1 and 9.1–10.5% on day 2. And after dose 4, fever was reported in 6.3–6.4% on day 1 and 7.3–9.7% on day 2.
Unsolicited Adverse Reactions in the Three US Infant and Toddler Safety Studies
The following were determined to be adverse drug reactions based on experience with Prevnar 13 in clinical trials.
Reactions occurring in greater than 1% of infants and toddlers: diarrhea, vomiting, and rash.
Reactions occurring in less than 1% of infants and toddlers: crying, hypersensitivity reaction (including face edema, dyspnea, and bronchospasm), seizures (including febrile seizures), and urticaria or urticaria-like rash.
Safety Assessments in the Catch-Up Studies in Infants and Children Through 5 Years of Age
In a catch-up study conducted in Poland (Study 4), 354 children (7 months through 5 years of age) receiving at least one dose of Prevnar 13 were also monitored for safety. All subjects in this study were White and non-Hispanic. Overall, 49.6% of subjects were male infants. The incidence and severity of solicited adverse reactions that occurred within 4 days following each dose of Prevnar 13 administered to pneumococcal-vaccine naïve children 7 months through 5 years of age are shown in Tables 5 and 6.
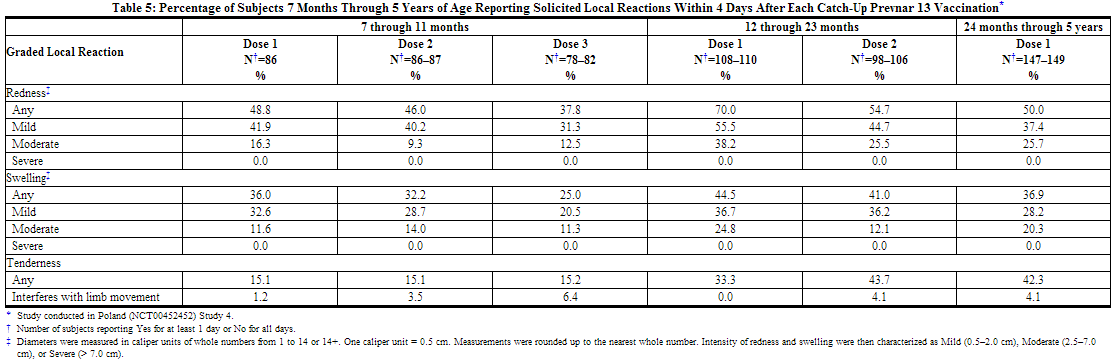
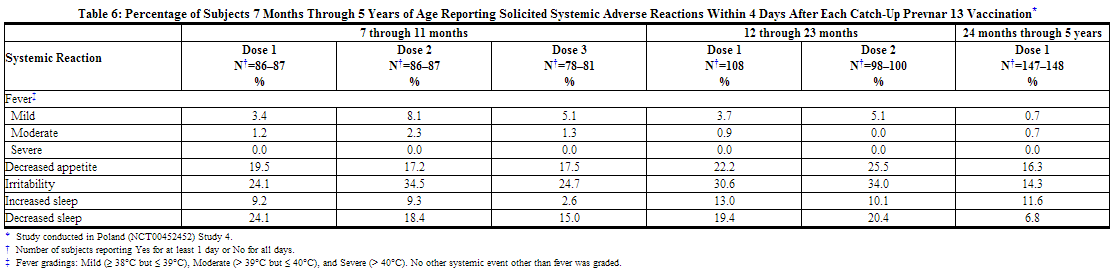
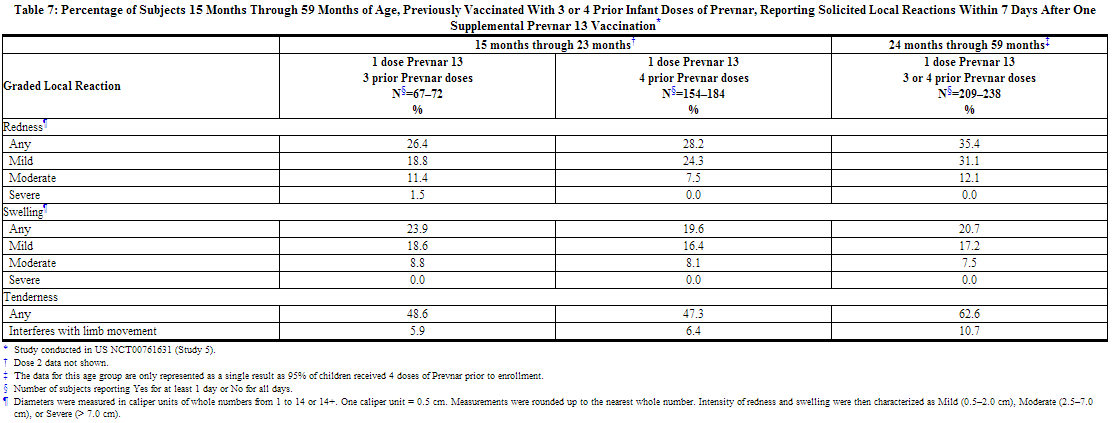
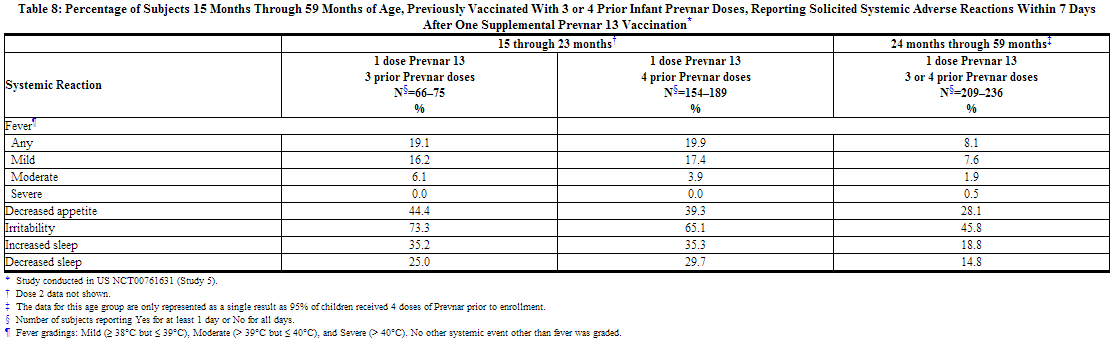
A US study (Study 5) evaluated the use of Prevnar 13 in children previously immunized with Prevnar. In this open label trial, 596 healthy children 15 through 59 months of age previously vaccinated with at least 3 doses of Prevnar, received 1 or 2 doses of Prevnar 13. Children 15 months through 23 months of age (group 1) received 2 doses, and children 24 months through 59 months of age (group 2) received one dose. Most subjects were White (74.3%), 14.9% were Black or African-American, and 1.2% were Asian; 89.3% of subjects were non-Hispanic and non-Latino and 10.7% were Hispanic or Latino. Overall, 52.2% of subjects were male. The incidence and severity of solicited adverse reactions that occurred within 7 days following one dose of Prevnar 13 administered to children 15 months through 59 months of age are shown in Tables 7 and 8.
Clinical Trials Experience With Prevnar 13 in Children 5 Through 17 Years of Age
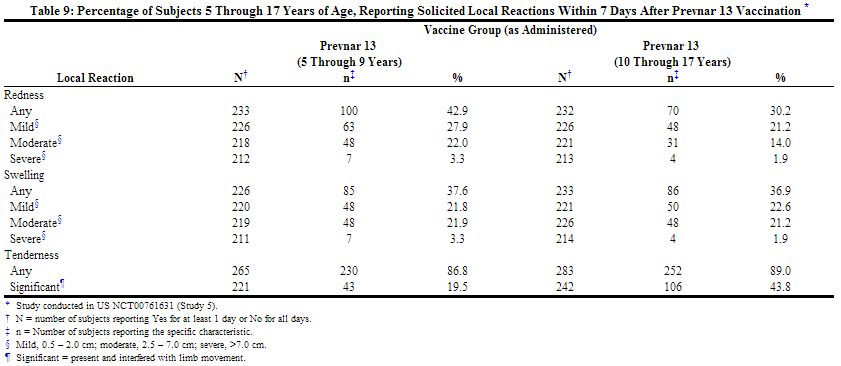
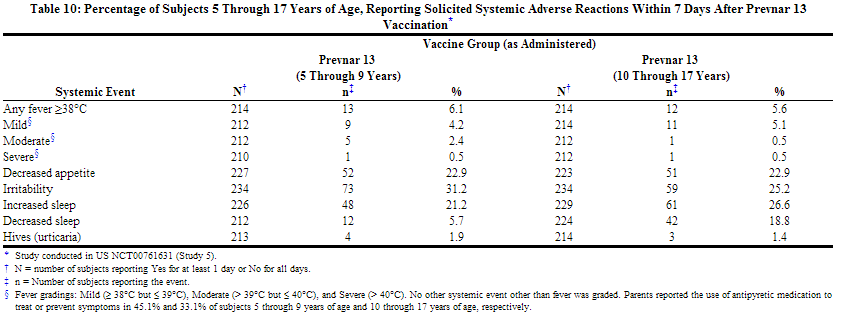
In a US study (Study 5), the safety of Prevnar 13 was evaluated in children 5 through 9 years of age previously immunized with at least one dose of Prevnar, and in children 10 through 17 years of age with no prior pneumococcal vaccination. In this open label trial, 592 children, including those with asthma, received a single dose of Prevnar 13. The percentage of children 5 through 9 years of age who received 3 and 4 prior doses of Prevnar was 29.1% and 54.5% respectively.
Most subjects were White (72.8%), 21.8% were Black or African-American, and 1.5% were Asian; 91.4% of subjects were non-Hispanic and non-Latino and 8.6% were Hispanic or Latino. Overall, 51.2% of subjects were male.
The incidence and severity of solicited adverse reactions that occurred within 7 days following one dose of Prevnar 13 administered to children 5 through 17 years of age are shown in Tables 9 and 10.
Clinical Trials Experience With Prevnar 13 in Adults ≥ 50 Years of Age
The safety of Prevnar 13 was assessed in 6 clinical studies conducted in the US and Europe which included 6,198 adults (5,667 received Prevnar 13) ranging in age from 50 through 95 years.
The 5,667 Prevnar 13 recipients included 2,616 adults who were aged 50 through 64 years and 3,051 adults aged 65 years and older. Of the 5,667 Prevnar 13 recipients, 3,751 adults had not previously received PPSV23 ("PPSV23 unvaccinated") and 1,916 adults were previously vaccinated ("PPSV23 previously vaccinated") with PPSV23 at least 3 years prior to enrollment.
Two of the 6 clinical studies supporting safety were randomized comparing the safety and immunogenicity of Prevnar 13 with PPSV23 as a single dose in PPSV23 unvaccinated adults aged 50 through 64 years (Study 6) and in adults ≥ 70 years PPSV23 previously vaccinated (≥ 5 years prior to enrollment) (Study 7). One study was randomized comparing the safety and immunogenicity of a single dose of Prevnar 13 compared to a single dose of PPSV23 in PPSV23 unvaccinated adults aged 60 through 64 years (Study 8). One clinical safety study (Study 9) of Prevnar 13, conducted in PPSV23 previously vaccinated (≥ 3 years prior to enrollment) adults aged ≥ 68 years was a single arm study. Two studies, one in the US (Study 10) in adults age 50 through 59 years and the other in Europe (Study 11) in adults aged ≥ 65 years, evaluated the concomitant administration of Prevnar 13 with trivalent inactivated influenza vaccine (Fluarix®, A/H1N1, A/H3N2, and B, Fall 2007/Spring 2008: TIV) in these two age groups in PPSV23 unvaccinated adults.
The total safety population in the 6 studies was 6,198. In 5 of the 6 studies, more females than males were enrolled (50.2% – 61.8%). Across the 6 studies the racial distribution included: > 91% White; 0.2%–7.5% Black or African American; 0%–1.7% Asian; < 1% Native Hawaiian or other Pacific Islander; ≤ 1%, American Indian or Alaskan Native. Ethnicity data were not collected in study 6; in the 5 other studies 0.6%–4.8% were Hispanic or Latino.
In five studies, persons with pre-existing underlying diseases were enrolled if the medical condition was stable (did not require a change in therapy or hospitalization for worsening disease for 12 weeks before receipt of study vaccine) except in study 9 where subjects were enrolled if the medical condition was stable for 6 or more weeks before receipt of study vaccine.
Persons were excluded from study participation due to prior receipt of diphtheria toxoid containing vaccines within 6 months of study vaccine. However, the time of prior receipt of a diphtheria toxoid containing vaccine was not recorded.
Solicited adverse reactions for Prevnar 13 were monitored by subjects recording local adverse reactions and systemic reactions daily using an electronic diary for 14 consecutive days following vaccination. Unsolicited serious and non-serious adverse events were collected for one month after each vaccination. In addition, serious adverse events were collected for an additional 5 months after each vaccination (at the 6-month follow-up phone contact) in all studies except Study 11.
Serious Adverse Events in Adult Clinical Studies
Across the 6 studies, serious adverse events within 1 month of vaccination were reported after an initial study dose in 0.2%–1.4% of 5055 persons vaccinated with Prevnar 13 and in 0.4%–1.7% of 1124 persons vaccinated after an initial study dose of PPSV23. From 1 month to 6 months after an initial study dose, serious adverse events were reported in 1.2%–5.8% of persons vaccinated during the studies with Prevnar 13 and in 2.4%–5.5% of persons vaccinated with PPSV23. One case of erythema multiforme occurred 34 days after receipt of a second dose of Prevnar 13.
Twelve of 5,667 (0.21%) Prevnar 13 recipients and 4 of 1,391 (0.29 %) PPSV23 recipients died. Deaths occurred between day 3 and day 309 after study vaccination with Prevnar 13 or PPSV23. Two of 12 deaths occurred within 30 days of vaccination and both deaths were in subjects > 65 years of age. One death due to cardiac failure occurred 3 days after receiving placebo. This subject had received Prevnar 13 and TIV one month earlier. The other death was due to peritonitis 20 days after receiving Prevnar 13. The reported causes of the 10 remaining deaths occurring greater than 30 days after receiving Prevnar 13 were cardiac disorders (4), neoplasms (4), Mycobacterium avium complex pulmonary infection (1) and septic shock (1).
Solicited Adverse Reactions in Adult Clinical Studies
The incidence and severity of solicited adverse reactions that occurred within 14 days following each dose of Prevnar 13 or PPSV23 administered to adults in 4 studies are shown in Tables 11, 12, 13, and 14. The commonly reported local adverse reactions after Prevnar 13 vaccination in PPSV23 unvaccinated and PPSV23 previously vaccinated adults were redness, swelling and pain at the injection site, or limitation of arm movement (Tables 11 and 12). The commonly reported systemic adverse reactions in PPSV23 unvaccinated and PPSV23 previously vaccinated adults were fatigue, headache, chills, rash, decreased appetite, or muscle pain and joint pain (Tables 13 and 14).
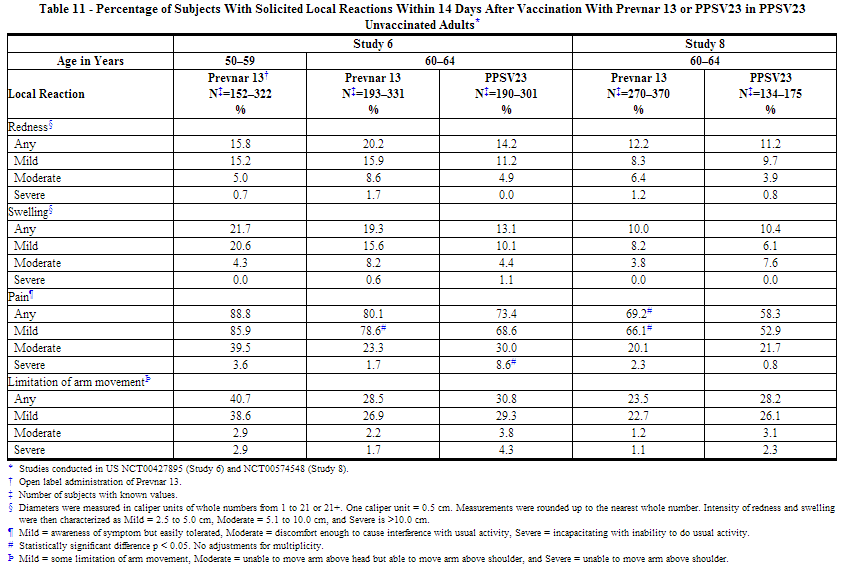
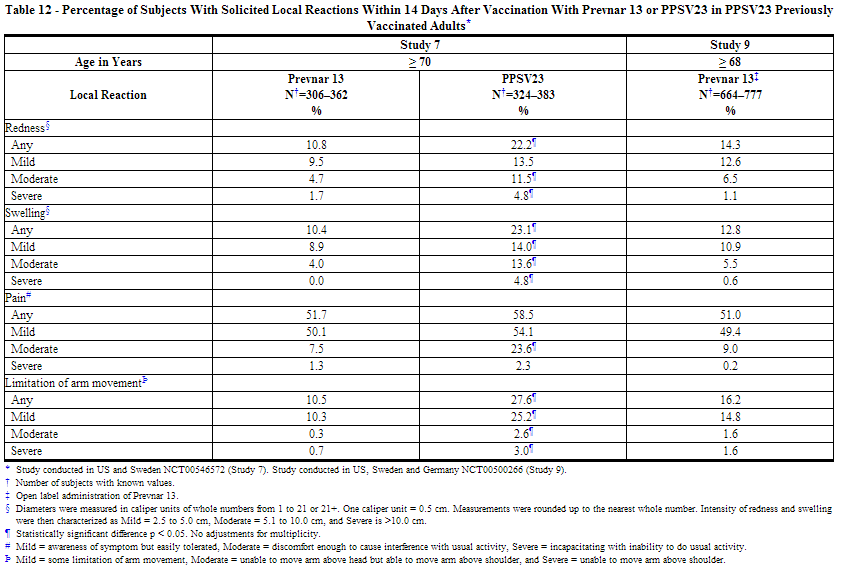
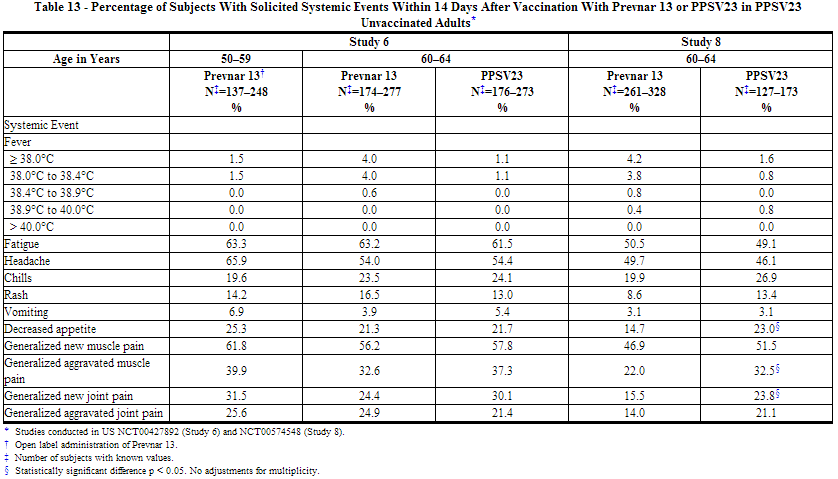
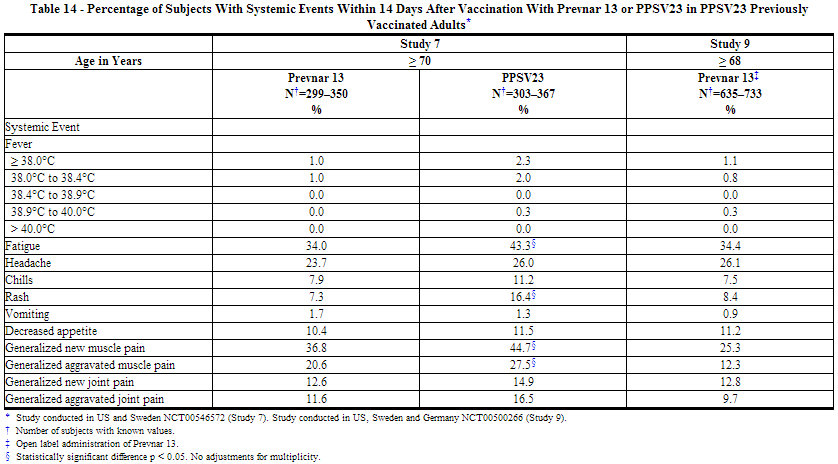
Solicited Adverse Reactions in Adult Clinical Studies of Concomitant Administration of Prevnar 13 and TIV (Fluarix)
The safety of concomitant administration of Prevnar 13 with TIV was assessed in 2 studies in PPSV23 unvaccinated adults aged 50 through 59 years (Study 10) and aged ≥ 65 years (Study 11).
Frequencies of local reactions within 14 days postvaccination in adults aged 50 through 59 years and in adults aged ≥ 65 years were similar after Prevnar 13 was administered with TIV compared to Prevnar 13 administered alone, with the exception of mild redness at the injection site, which was increased when Prevnar 13 was administered concomitantly with TIV and mild limitation of arm movement, which was increased when Prevnar 13 was administered alone.
An increase in some solicited systemic reactions within 14 days postvaccination was noted when Prevnar 13 was administered concomitantly with TIV compared with TIV given alone (headache, chills, rash, decreased appetite, muscle and joint pain) or with Prevnar 13 given alone (fatigue, headache, chills, decreased appetite, and joint pain).
Clinical Trials Experience With Prevnar in Infants and Toddlers
The safety experience with Prevnar is relevant to Prevnar 13 because the two vaccines share common components.
Generally, the adverse reactions reported in clinical trials with Prevnar 13 were also reported in clinical trials with Prevnar.
Overall, the safety of Prevnar was evaluated in a total of five clinical studies in the U.S. in which 18,168 infants and children received a total of 58,699 doses of vaccine at 2, 4, 6, and 12–15 months of age.
Adverse events reported in clinical trials with Prevnar that occurred within 3 days of vaccination in infants and toddlers and resulted in emergency room visits or hospitalizations, but were not presented in Section 6.1 as adverse reactions for Prevnar 13 are listed below:
- Bronchiolitis
- UTI
- Acute gastroenteritis
- Asthma
- Aspiration
- Breath holding
- Influenza
- Inguinal hernia repair
- Viral syndrome
- URI
- Croup
- Thrush
- Wheezing
- Choking
- Conjunctivitis
- Pharyngitis
- Colic
- Colitis
- Congestive heart failure
- Roseola
- Sepsis
Postmarketing Experience
Post-marketing Experience With Prevnar 13 in Infants and Toddlers
The following adverse events have been reported through passive surveillance since market introduction of Prevnar 13. Because these events are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to the vaccine. The following adverse events were included based on one or more of the following factors: severity, frequency of reporting, or strength of evidence for a causal relationship to Prevnar 13 vaccine.
- Administration site conditions: Vaccination-site dermatitis, vaccination-site pruritus, vaccination-site urticaria
- Blood and lymphatic system disorders: Lymphadenopathy localized to the region of the injection site
- Cardiac Disorders: Cyanosis
- Immune system disorders: Anaphylactic/anaphylactoid reaction including shock
- Nervous System Disorders: Hypotonia
- Skin and subcutaneous tissue disorders: Angioneurotic edema, erythema multiforme
- Respiratory: Apnea
- Vascular Disorders: Pallor
Drug Interactions
- Drug 1
- Drug 2
- Drug 3
- Drug 4
- Drug 5
Drug 1
(Description)
Drug 2
(Description)
Drug 3
(Description)
Drug 4
(Description)
Drug 5
(Description)
Use in Specific Populations
Pregnancy
Pregnancy Category (FDA):
(Description)
Pregnancy Category (AUS):
(Description)
Labor and Delivery
(Description)
Nursing Mothers
(Description)
Pediatric Use
(Description)
Geriatic Use
(Description)
Gender
(Description)
Race
(Description)
Renal Impairment
(Description)
Hepatic Impairment
(Description)
Females of Reproductive Potential and Males
(Description)
Immunocompromised Patients
(Description)
Others
(Description)
Administration and Monitoring
Administration
(Oral/Intravenous/etc)
Monitoring
Condition 1
(Description regarding monitoring, from Warnings section)
Condition 2
(Description regarding monitoring, from Warnings section)
Condition 3
(Description regarding monitoring, from Warnings section)
IV Compatibility
Solution
Compatible
- Solution 1
- Solution 2
- Solution 3
Not Tested
- Solution 1
- Solution 2
- Solution 3
Variable
- Solution 1
- Solution 2
- Solution 3
Incompatible
- Solution 1
- Solution 2
- Solution 3
Y-Site
Compatible
- Solution 1
- Solution 2
- Solution 3
Not Tested
- Solution 1
- Solution 2
- Solution 3
Variable
- Solution 1
- Solution 2
- Solution 3
Incompatible
- Solution 1
- Solution 2
- Solution 3
Admixture
Compatible
- Solution 1
- Solution 2
- Solution 3
Not Tested
- Solution 1
- Solution 2
- Solution 3
Variable
- Solution 1
- Solution 2
- Solution 3
Incompatible
- Solution 1
- Solution 2
- Solution 3
Syringe
Compatible
- Solution 1
- Solution 2
- Solution 3
Not Tested
- Solution 1
- Solution 2
- Solution 3
Variable
- Solution 1
- Solution 2
- Solution 3
Incompatible
- Solution 1
- Solution 2
- Solution 3
TPN/TNA
Compatible
- Solution 1
- Solution 2
- Solution 3
Not Tested
- Solution 1
- Solution 2
- Solution 3
Variable
- Solution 1
- Solution 2
- Solution 3
Incompatible
- Solution 1
- Solution 2
- Solution 3
Overdosage
Acute Overdose
Signs and Symptoms
(Description)
Management
(Description)
Chronic Overdose
Signs and Symptoms
(Description)
Management
(Description)
Pharmacology
Pneumococcal Vaccine 13-Valent
| |
| Systematic (IUPAC) name | |
| ? | |
| Identifiers | |
| CAS number | ? |
| ATC code | ? |
| PubChem | ? |
| Chemical data | |
| Formula | ? |
| Mol. mass | ? |
| Pharmacokinetic data | |
| Bioavailability | ? |
| Metabolism | ? |
| Half life | ? |
| Excretion | ? |
| Therapeutic considerations | |
| Pregnancy cat. |
? |
| Legal status | |
| Routes | ? |
Mechanism of Action
(Description)
Structure
(Description with picture)
Pharmacodynamics
(Description)
Pharmacokinetics
(Description)
Nonclinical Toxicology
(Description)
Clinical Studies
Condition 1
(Description)
Condition 2
(Description)
Condition 3
(Description)
How Supplied
(Description)
Storage
There is limited information regarding Pneumococcal Vaccine 13-Valent Storage in the drug label.
Images
Drug Images
{{#ask: Page Name::Pneumococcal Vaccine 13-Valent |?Pill Name |?Drug Name |?Pill Ingred |?Pill Imprint |?Pill Dosage |?Pill Color |?Pill Shape |?Pill Size (mm) |?Pill Scoring |?NDC |?Drug Author |format=template |template=DrugPageImages |mainlabel=- |sort=Pill Name }}
Package and Label Display Panel
{{#ask: Label Page::Pneumococcal Vaccine 13-Valent |?Label Name |format=template |template=DrugLabelImages |mainlabel=- |sort=Label Page }}
Patient Counseling Information
(Patient Counseling Information)
Precautions with Alcohol
Alcohol-Pneumococcal Vaccine 13-Valent interaction has not been established. Talk to your doctor about the effects of taking alcohol with this medication.
Brand Names
There is limited information regarding Pneumococcal Vaccine 13-Valent Brand Names in the drug label.
Look-Alike Drug Names
- (Paired Confused Name 1a) — (Paired Confused Name 1b)
- (Paired Confused Name 2a) — (Paired Confused Name 2b)
- (Paired Confused Name 3a) — (Paired Confused Name 3b)
Drug Shortage Status
Drug Shortage
Price
References
The contents of this FDA label are provided by the National Library of Medicine.