Cirrhosis pathophysiology
| https://https://www.youtube.com/watch?v=5szNmKtyBW4%7C350}} |
|
Cirrhosis Microchapters |
|
Diagnosis |
|---|
|
Treatment |
|
Case studies |
|
Cirrhosis pathophysiology On the Web |
|
American Roentgen Ray Society Images of Cirrhosis pathophysiology |
|
Risk calculators and risk factors for Cirrhosis pathophysiology |
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1] Associate Editor(s)-in-Chief: Aditya Govindavarjhulla, M.B.B.S. [2];Kalsang Dolma, M.B.B.S.[3]
Please help WikiDoc by adding content here. It's easy! Click here to learn about editing.
Overview
Cirrhosis occurs due to long term liver injury which causes an imbalance between matrix production and degradation. Early disruption of the normal hepatic matrix results in its replacement by scar tissue, which in turn has deleterious effects on cell function.
Pathophysiology
- The liver plays a vital role in the synthesis of proteins (e.g. albumin, clotting factors and complement), detoxification, and storage (e.g. vitamin A). In addition, it participates in the metabolism of lipids and carbohydrates.
- Cirrhosis is often preceded by hepatitis and fatty liver (steatosis). If the cause is removed at this stage, the changes are still fully reversible.
- The pathological hallmark of cirrhosis is the development of scar tissue that replaces normal parenchyma, blocking the portal flow of blood through the organ and disturbing normal function. The development of fibrosis requires several months, or even years, of ongoing injury.
- Recent research shows the pivotal role of the stellate cell, a cell type that normally stores vitamin A, in the development of cirrhosis. Damage to the hepatic parenchyma leads to the activation of the stellate cell, which becomes contractile (called myofibroblast) and obstructs blood flow in the circulation. In addition, it secretes TGF-β1, which leads to a fibrotic response and proliferation of connective tissue. The extracellular matrix around hepatocytes is composed of collagens (especially type I, III, IV), glycoprotein and proteoglycans.
- Sinusoidal endothelial cells are also important contributors of early fibrosis. Endothelial cells from a normal liver produces collagen, laminin and fibronectin.[1][2]
- In addition, the liver responds to injury with new blood vessel formation. Platelet derived growth factor (PDGF), vascular endothelial growth factor (VEGF), nitric oxide, and carbon monoxide are the mediators involved in angiogenesis. Angiogenesis in cirrhosis results in the production of immature and permeable VEGF induced neo-vessels that fail to correct liver injury. [3],[4]
- Furthermore, it disturbs the balance between matrix metalloproteinases and the naturally occurring inhibitors (TIMP 1 and 2), leading tomatrix breakdown and replacement by connective tissue-secreted matrix.[5]. Matrix metalloproteinase (MMP) are calcium dependent enzymes that specifically degrade collagen and non collagenous substrate. There are five categories of MMP based upon their specificity for the substrate. MMP-2 and stromyelysin-1 are produced from stellate cells. MMP-2 degrades collagen and stromelysin-1 degrades proteoglycan and glycoprotein.
- The fibrous tissue bands (septa) separate hepatocyte nodules, which eventually replace the entire liver architecture, leading to decreased blood flow throughout.
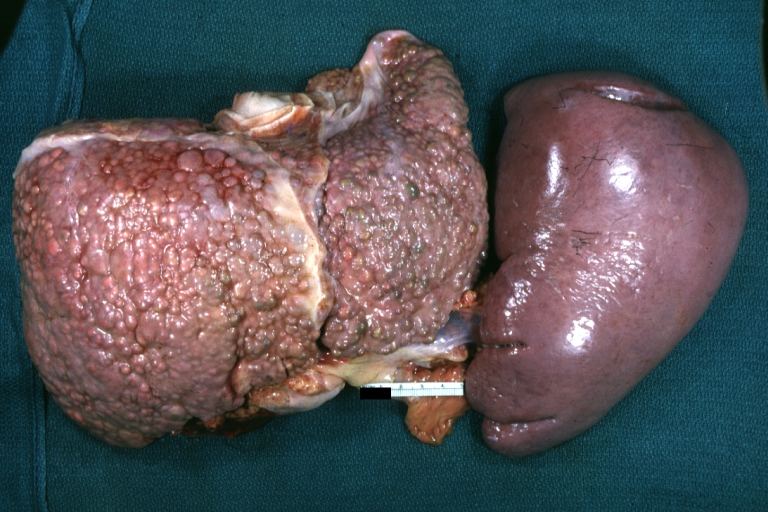
- The spleen becomes congested, which leads to hypersplenism and increased sequestration of platelets.
- Portal hypertension is responsible for the most severe complications of cirrhosis.
- Pathogenesis of cirrhosis based upon its individual cause is as follows:
- Alcoholic liver disease: Alcohol seems to injure the liver by blocking the normal metabolism of protein, fats, and carbohydrates. Patients may also have concurrent alcoholic hepatitis with fever, hepatomegaly, jaundice, and [[anorexia].
- Chronic hepatitis C: Infection with the hepatitis C virus causes inflammation of and low grade damage to the liver that over several decades can lead to cirrhosis.
- Non-alcoholic steatohepatitis (NASH): In NASH, fat builds up in the liver and eventually causes scar tissue. This type of hepatitis appears to be associated with diabetes, protein malnutrition, obesity, coronary artery disease, and treatment with corticosteroid medications.
- Primary sclerosing cholangitis: PSC is a progressive cholestatic disorder presenting with pruritus, steatorrhea, fat soluble vitamin deficiencies, and metabolic bone disease. There is a strong association with inflammatory bowel disease (IBD), especially ulcerative colitis.
- Autoimmune hepatitis: This disease is caused by the immunologic damage to the liver causing inflammation and eventually scarring and cirrhosis.
Genetics
- Certain TERT (Telomerase reverese transcriptase)gene variants resulting in reduced telomerase activity has been found to be a risk factor for sporadic cirrhosis[6]
- An uncharacterized nucleolar protein, NOL11, has a role in the pathogenesis of North American Indian childhood cirrhosis[7]
- Loss of interaction between the C-terminus of Utp4/cirhin and other SSU processome proteins may cause North American Indian childhood cirrhosis[8]
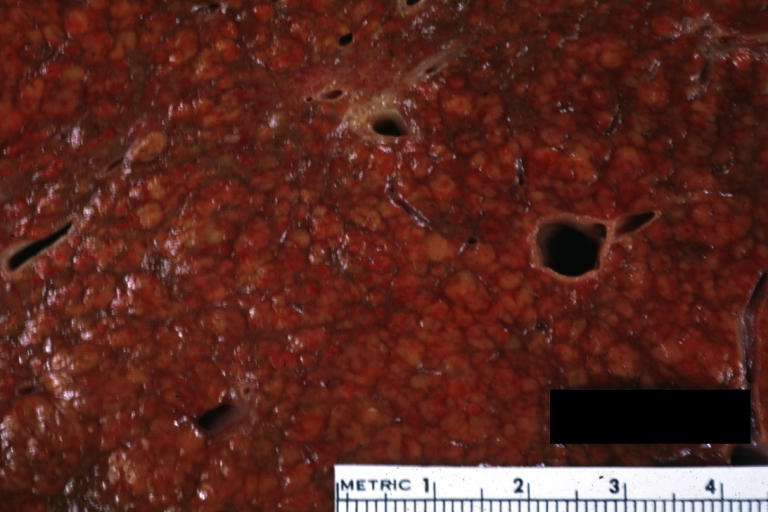
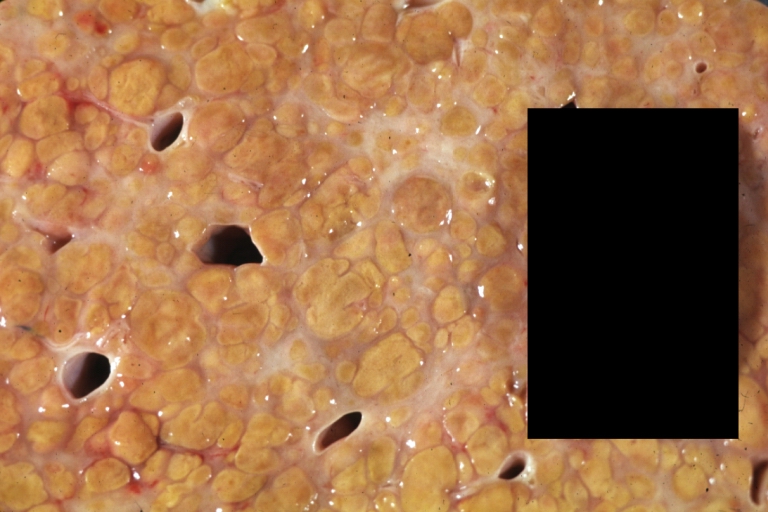
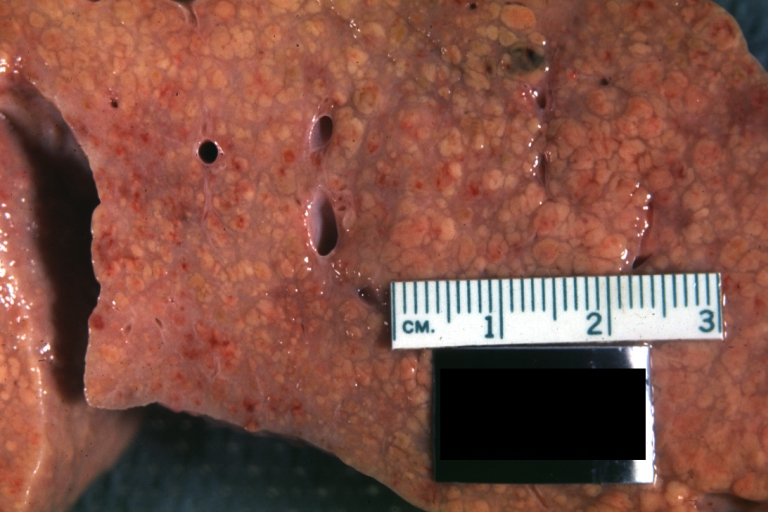
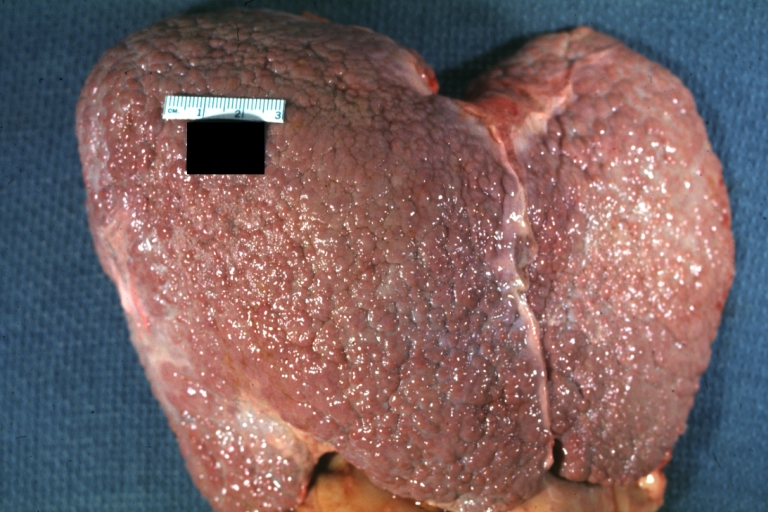
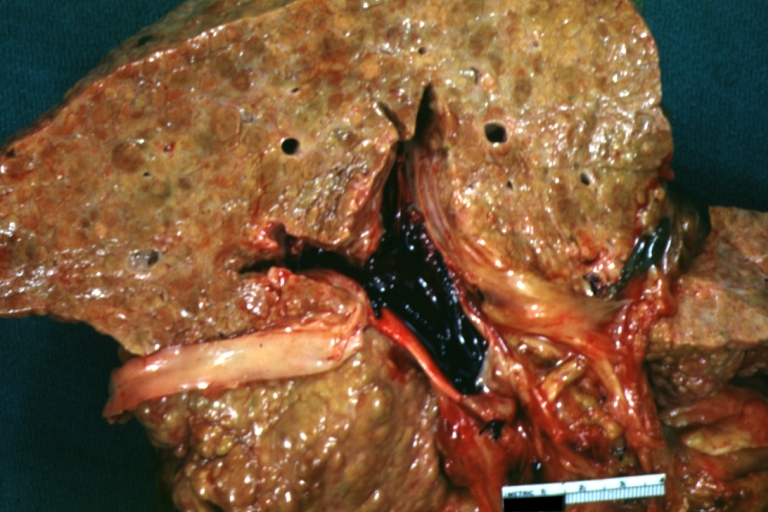
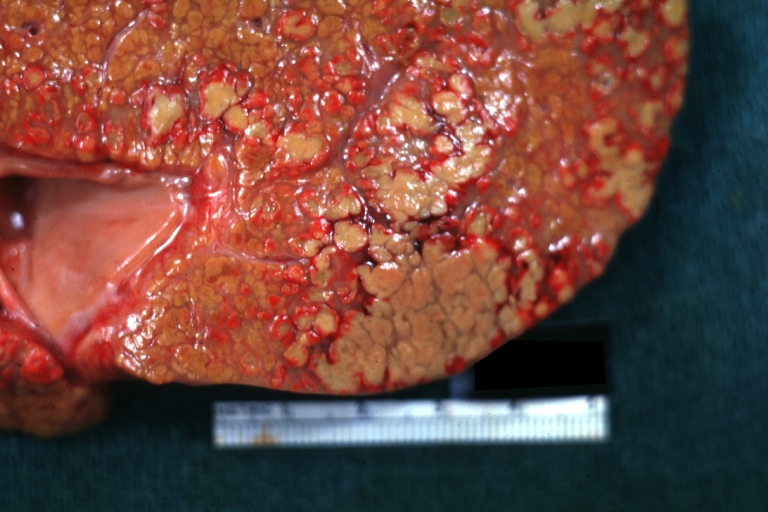
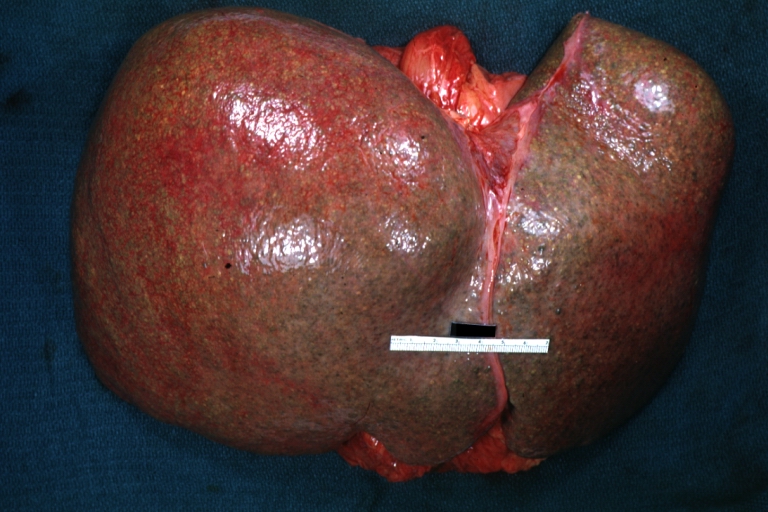
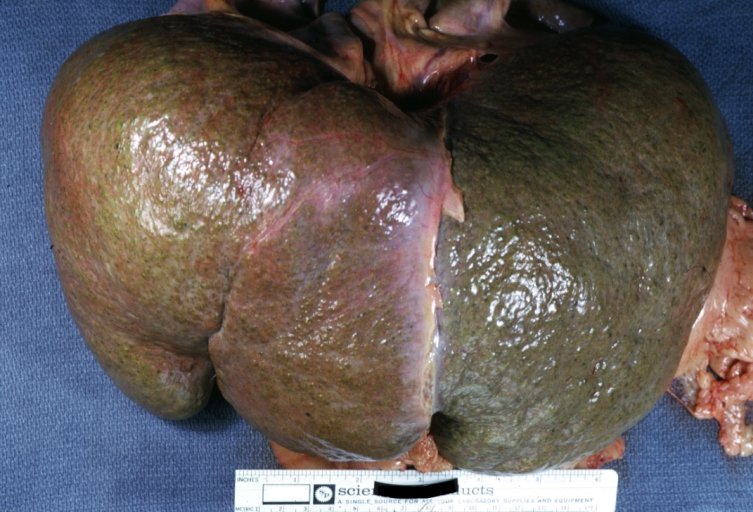
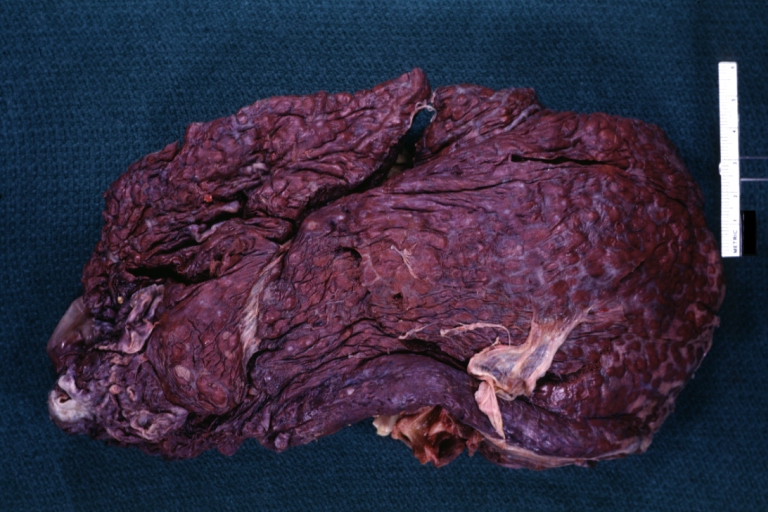
Gross Pathology
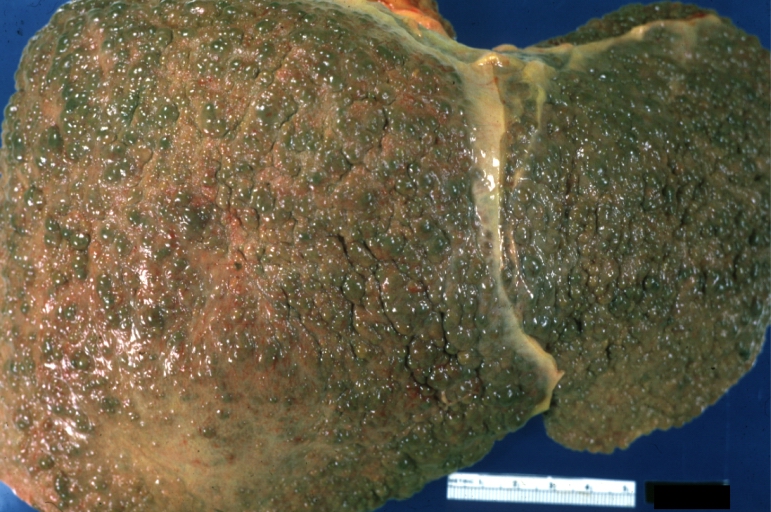
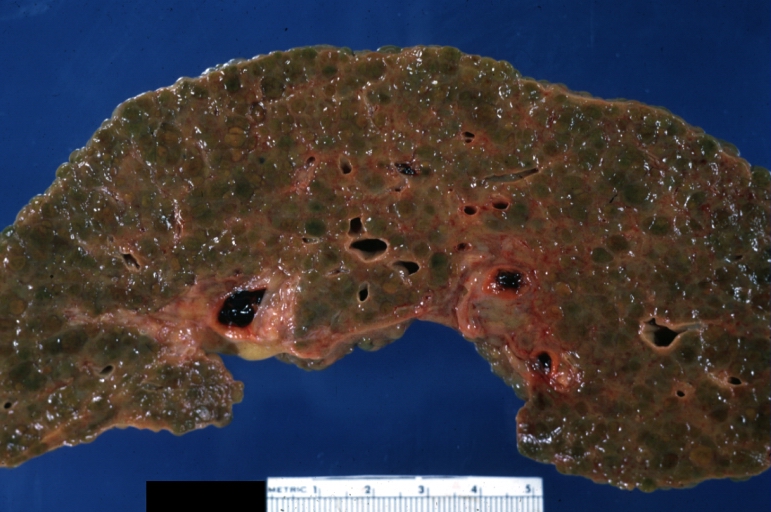
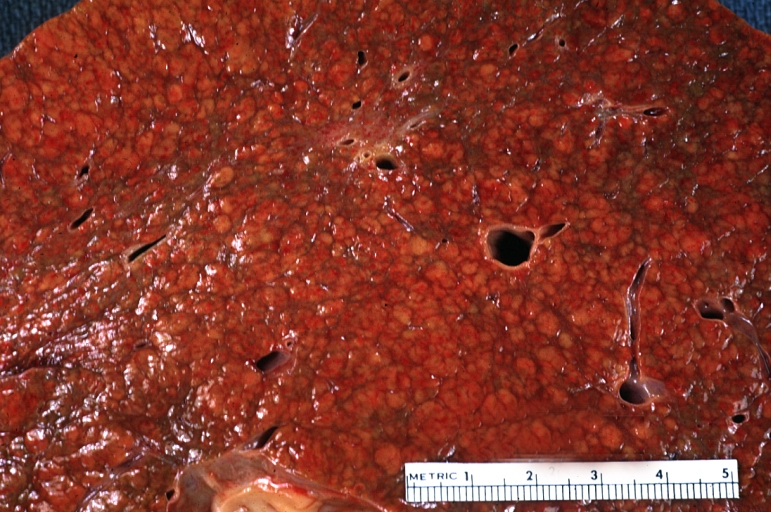
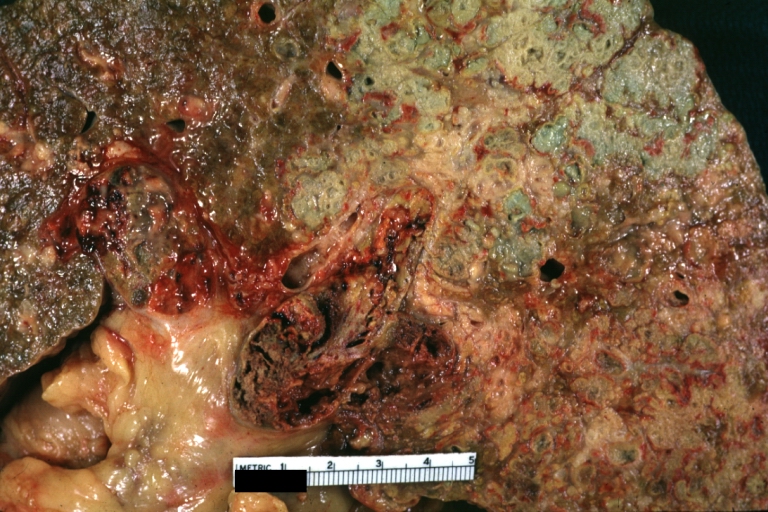
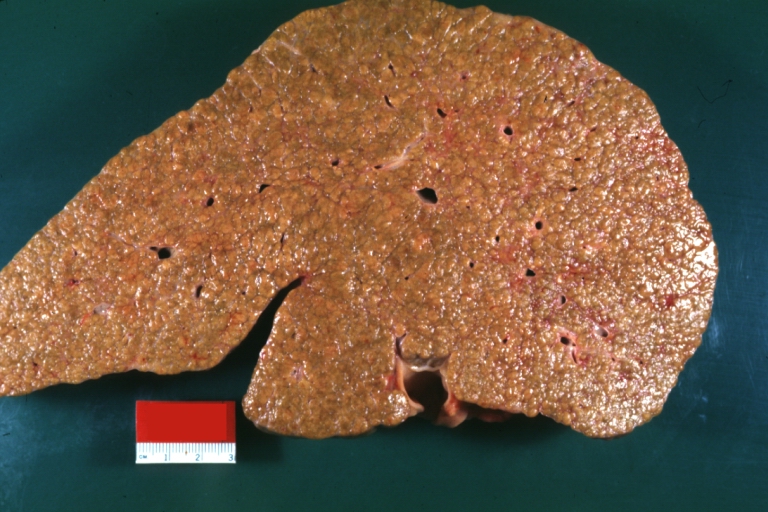
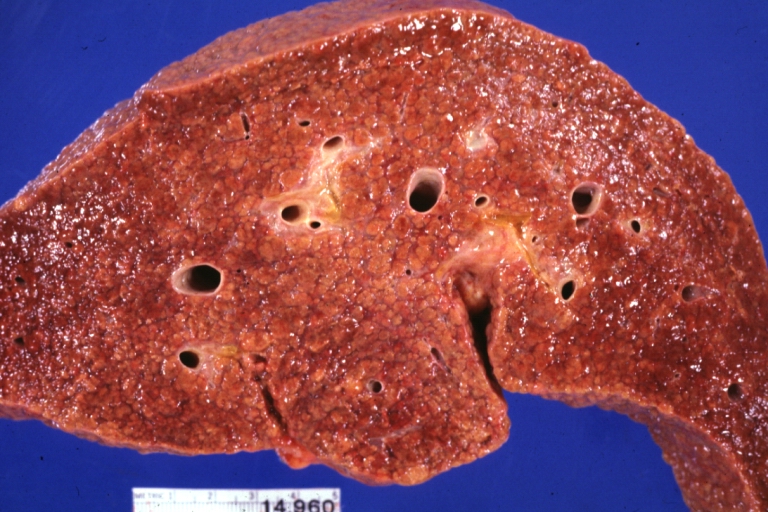
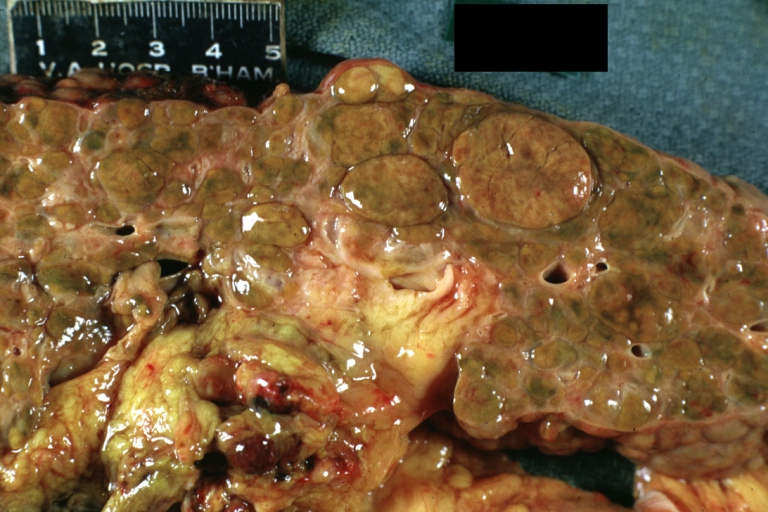
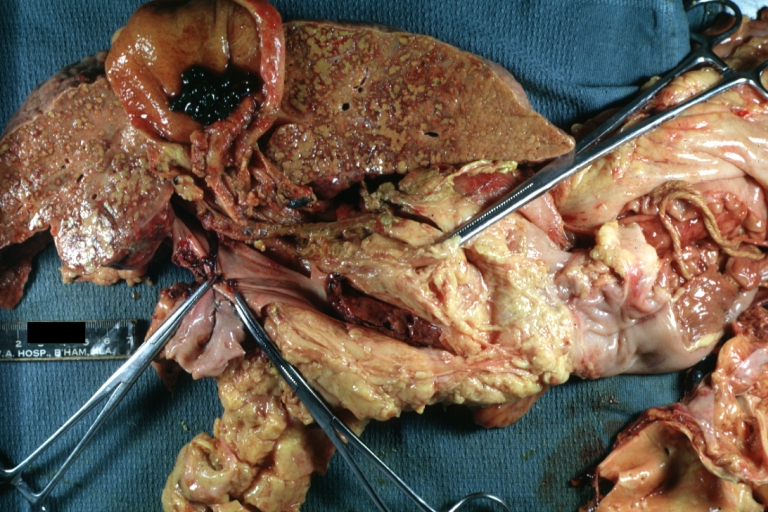
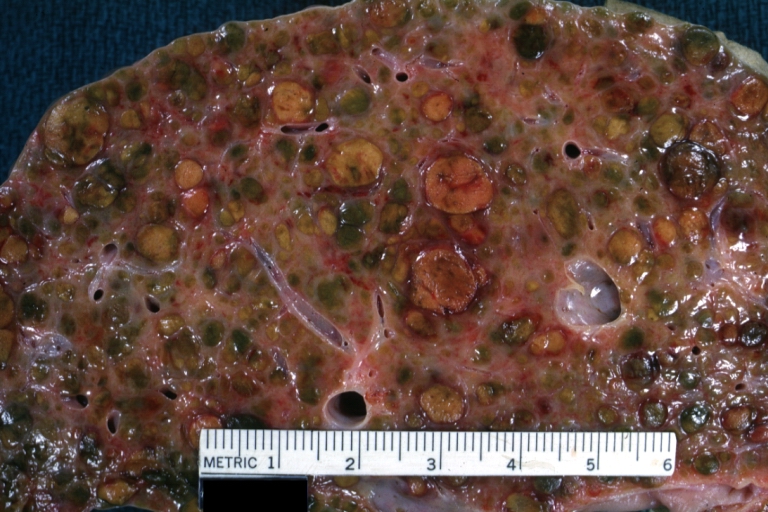
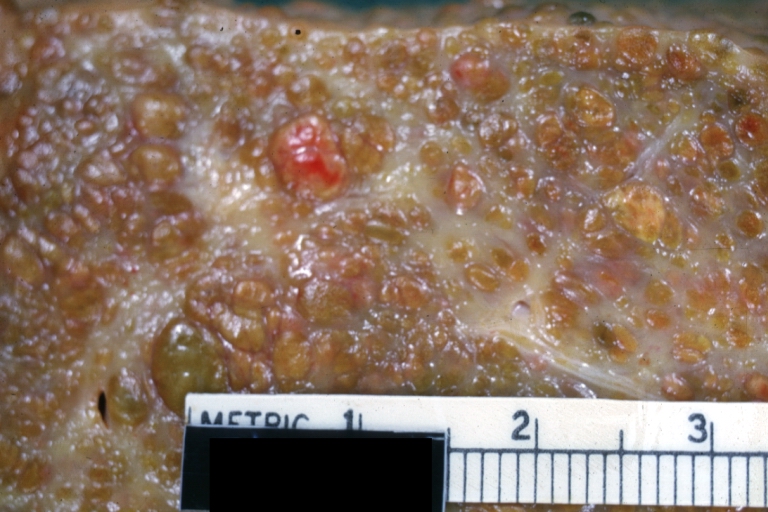
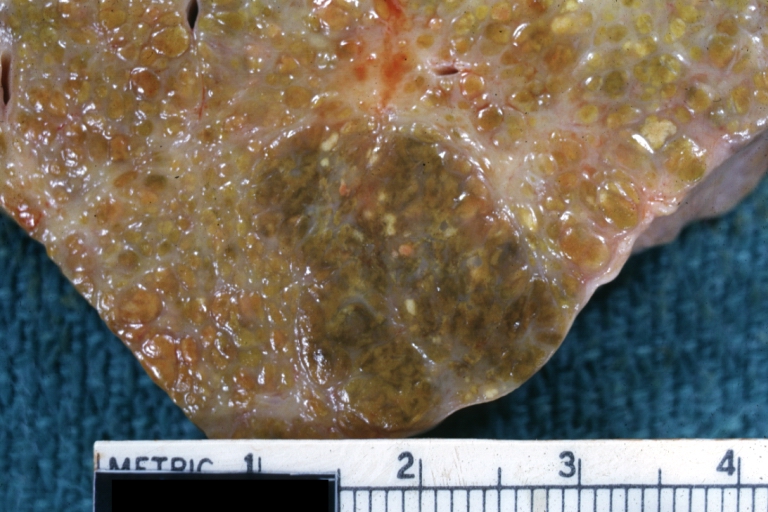
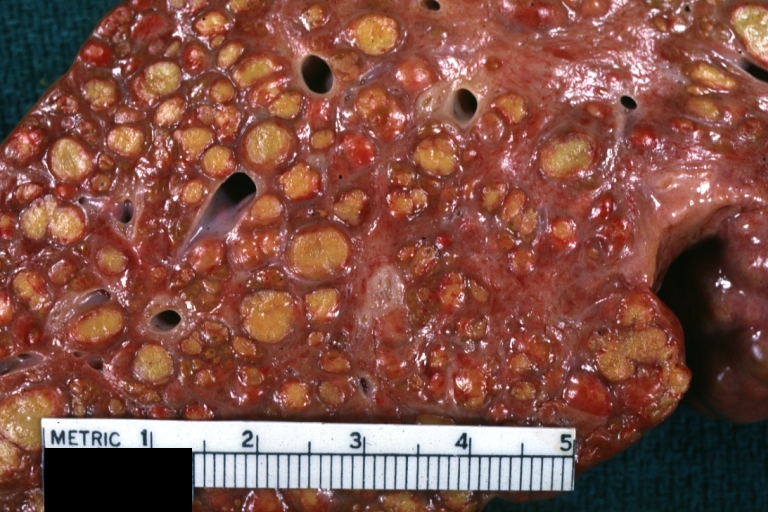
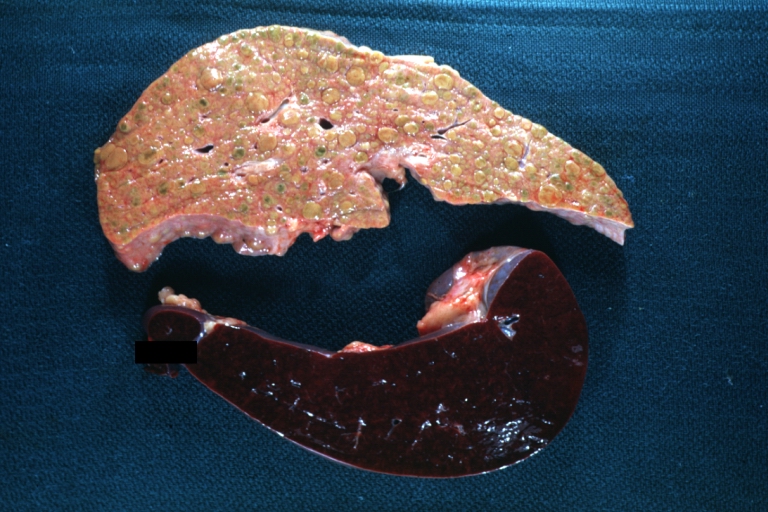
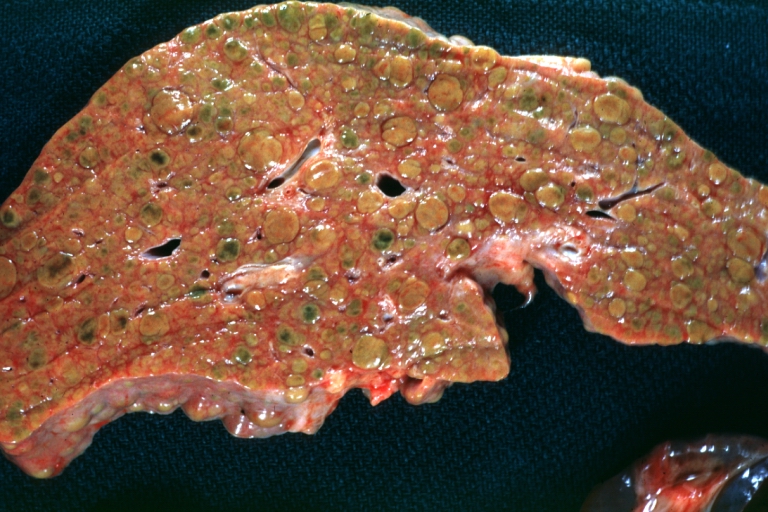
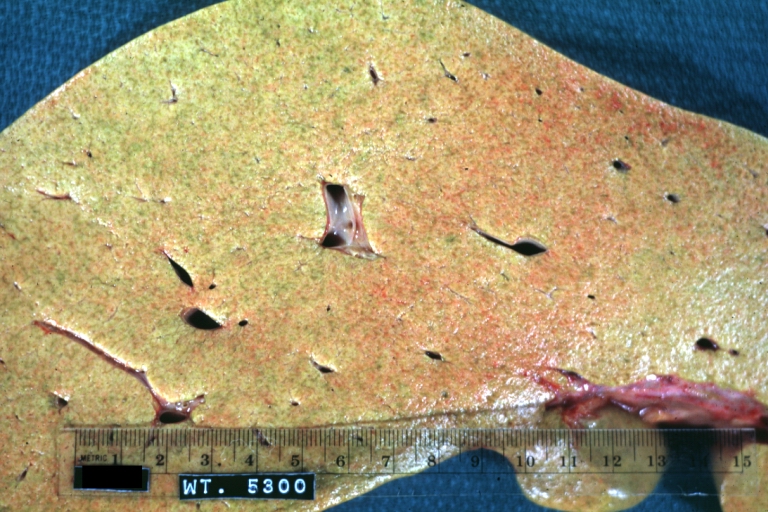
Macroscopically, the liver may initially be enlarged, but with progression of the disease, it becomes smaller. Its surface is irregular, the consistency is firm, and the color is often yellow (if associates steatosis). Depending on the size of the nodules there are three macroscopic types: micronodular, macronodular and mixed cirrhosis.
- In the micronodular form (Laennec's cirrhosis or portal cirrhosis) regenerating nodules are under 3 mm.
- In macronodular cirrhosis (post-necrotic cirrhosis), the nodules are larger than 3 mm.
- The mixed cirrhosis consists of a variety of nodules with different sizes.
-
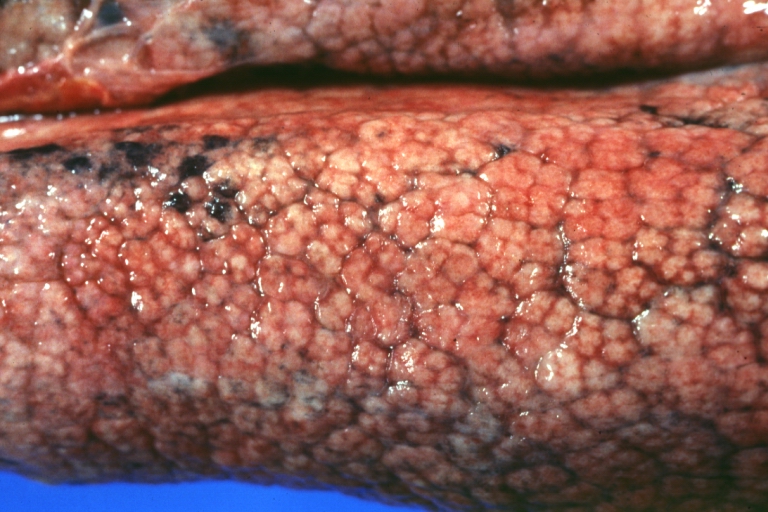
Cirrhosis: Gross, external view of micronodular cirrhosis
-
Cirrhosis: Gross, cut section of previous one (an excellent example)
-
Cirrhosis: Gross, close-up image
-
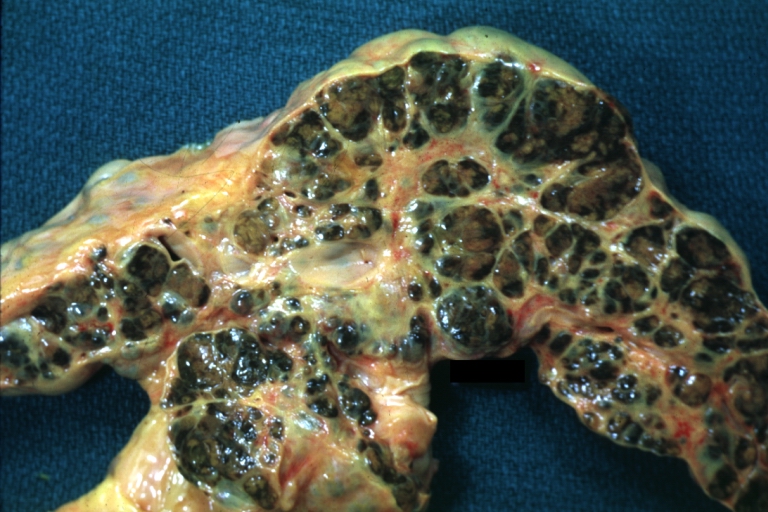
Macronodular cirrhosis and hepatoma
-
Cirrhosis: Gross, close-up, natural color (an excellent example)
-
Cirrhosis: Gross, close-up (an excellent example)
-
Cirrhosis: Gross, close-up view
-
Micronodular cirrhosis: Gross, external view (an excellent example)
-
Micronodular cirrhosis: Gross, close-up image
-
Micronodular cirrhosis: Gross (an excellent example)
-
Macronodular cirrhosis: Gross, natural color (perfect color for cirrhosis), close-up, an excellent example
-
Cirrhosis with portocaval shunt: Gross, severe cirrhosis with extensive liver necrosis due to thrombosis of portocaval shunt (well shown)
-
Endstage cirrhosis: Gross, natural color, close-up (an excellent example)
-
Endstage cirrhosis: Gross, natural color, close-up view is an excellent example for nodules of yellow-orange liver tissue and broad irregular bands of fibrosis
-
Endstage cirrhosis: Gross, natural color, close-up cut surface, very well shown nodules of yellow and necrotic opaque liver tissue with broad and irregular bands of fibrosis (an excellent example)
-
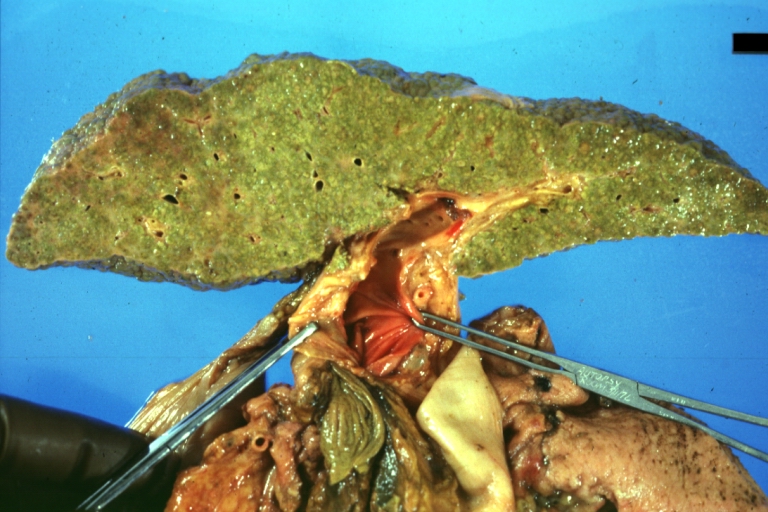
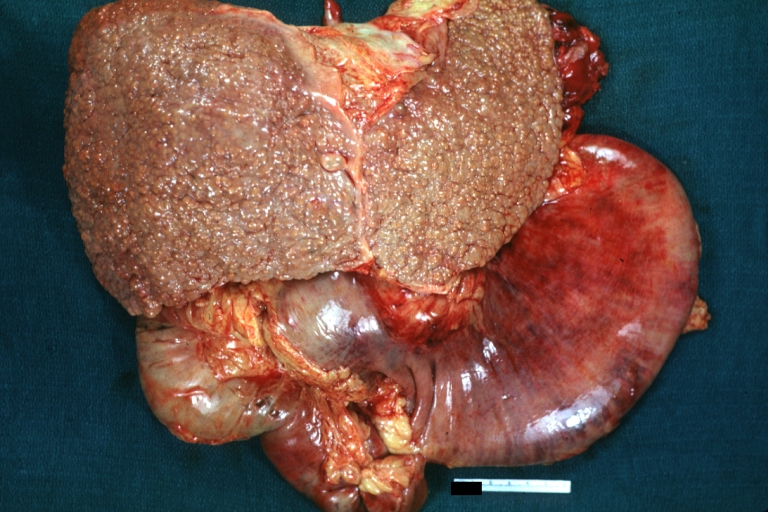
Macronodular cirrhosis: Gross, natural color, external view of liver and very enlarged spleen (liver has variable size nodules up to about 2 cm)
-
Macronodular cirrhosis: Gross, natural color, cut surface, large irregular bands of fibrosis with variable size liver cell nodules up to about 8 mm and all necrotic appears to be an end stage liver disease.
-
Macronodular cirrhosis: Gross, natural color view of frontal sections of liver and spleen showing a contracted macronodular liver and an enlarged spleen as large as the liver
-
Macronodular cirrhosis: Gross, natural color slab of liver
-
Fatty change and early cirrhosis: Gross, natural color, rather close-up image showing typical fatty color, and in lighting at lower right of micrography micronodularity is evident (quite good example)
-
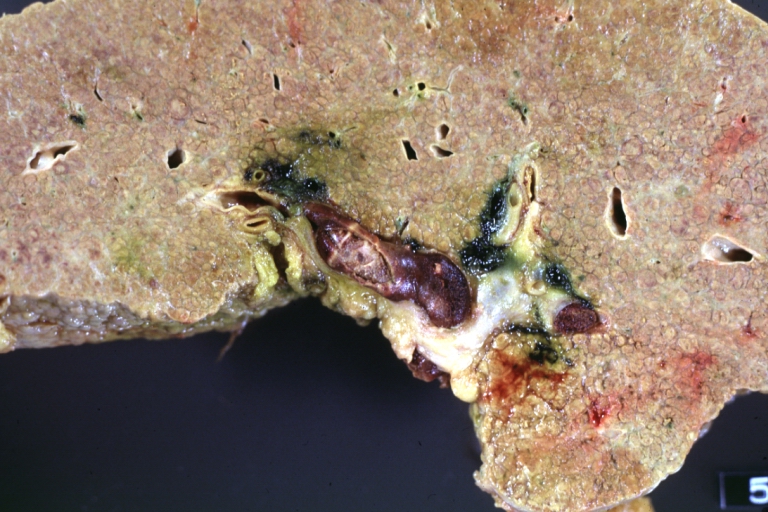
Cirrhosis with portal vein thrombosis: Gross, natural color, sectioned liver with portal vein exposed and filled with red thrombus. A good example of end stage cirrhosis.
-
Endstage cirrhosis with lobular necrosis: Gross, natural color, very close-up view (an excellent example of alcoholic cirrhosis)
-
Micronodular cirrhosis: Gross, natural color view of whole liver through capsule with obvious cirrhosis (note to quite large liver)
-
Micronodular cirrhosis: Gross, natural color, view of whole liver showing external surface typical cirrhotic liver (history of alcoholism)
-
Lung: Idiopathic Interstitial Fibrosis: Gross, natural color, an excellent photo of lung cirrhosis (close-up view)
-
Endstage cirrhosis: Gross, natural color, slice of liver. Portal vein is opened to show size and patency.
-
Endstage cirrhosis: Gross, natural color, severe cirrhosis with bile stasis
-
Portal Vein Thrombosis with cirrhosis: Gross, close-up, micronodular cirrhosis with portal vein thrombosis
-
Lung: Hematite: Gross, natural color, external view of "pulmonary cirrhosis" with typical hematite color
-
Gross, natural color of liver and stomach view from external surfaces, micronodular cirrhosis and hemorrhagic gastritis (as the surgeon would see these in natural color)
Microscopic Pathology
Microscopically, cirrhosis is characterized by regeneration nodules surrounded by fibrous septa. In these nodules, regenerating hepatocytes are disorderly disposed. Portal tracts, central veins and the radial pattern of hepatocytes are absent. Fibrous septa are important and may present inflammatory infiltrate (lymphocytes, macrophages). If it is a secondary biliary cirrhosis, biliary ducts are damaged, proliferated or distended - bile stasis. These dilated ducts contain inspissated bile which appears as bile casts or bile thrombi (brown-green, amorphous). Bile retention may be found also in the parenchyma, as the so called "bile lakes".[9]
Chronic active hepatitis - Cirrhosis
{{#ev:youtube|CzKGvWZrUpU}}
Micronodular cirrhosis
{{#ev:youtube|CV8OYeIUXko}}
Primary biliary cirrhosis
{{#ev:youtube|Jj8ozr_IttM}}
References
- ↑ Maher JJ, McGuire RF (1990). "Extracellular matrix gene expression increases preferentially in rat lipocytes and sinusoidal endothelial cells during hepatic fibrosis in vivo". J. Clin. Invest. 86 (5): 1641–8. doi:10.1172/JCI114886. PMC 296914. PMID 2243137. Unknown parameter
|month=ignored (help) - ↑ Herbst H, Frey A, Heinrichs O; et al. (1997). "Heterogeneity of liver cells expressing procollagen types I and IV in vivo". Histochem. Cell Biol. 107 (5): 399–409. PMID 9208331. Unknown parameter
|month=ignored (help) - ↑ Lee JS, Semela D, Iredale J, Shah VH (2007). "Sinusoidal remodeling and angiogenesis: a new function for the liver-specific pericyte?". Hepatology. 45 (3): 817–25. doi:10.1002/hep.21564. PMID 17326208. Unknown parameter
|month=ignored (help) - ↑ Rosmorduc O, Housset C (2010). "Hypoxia: a link between fibrogenesis, angiogenesis, and carcinogenesis in liver disease". Semin. Liver Dis. 30 (3): 258–70. doi:10.1055/s-0030-1255355. PMID 20665378. Unknown parameter
|month=ignored (help) - ↑ Iredale JP. Cirrhosis: new research provides a basis for rational and targeted treatments. BMJ 2003;327:143-7.Fulltext. PMID 12869458.
- ↑ Calado RT, Brudno J, Mehta P; et al. (2011). "Constitutional telomerase mutations are genetic risk factors for cirrhosis". Hepatology. 53 (5): 1600–7. doi:10.1002/hep.24173. PMC 3082730. PMID 21520173. Unknown parameter
|month=ignored (help) - ↑ Freed EF, Prieto JL, McCann KL, McStay B, Baserga SJ (2012). "NOL11, Implicated in the Pathogenesis of North American Indian Childhood Cirrhosis, Is Required for Pre-rRNA Transcription and Processing". PLoS Genet. 8 (8): e1002892. doi:10.1371/journal.pgen.1002892. PMC 3420923. PMID 22916032. Unknown parameter
|month=ignored (help) - ↑ Freed EF, Baserga SJ (2010). "The C-terminus of Utp4, mutated in childhood cirrhosis, is essential for ribosome biogenesis". Nucleic Acids Res. 38 (14): 4798–806. doi:10.1093/nar/gkq185. PMC 2919705. PMID 20385600. Unknown parameter
|month=ignored (help) - ↑ Pathology atlas, "cirrhosis".