Cefprozil microbiology
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1];Associate Editor(s)-in-Chief: Abdurahman Khalil, M.D. [2]
Cefprozil has in vitro activity against a broad range of gram-positive and gram-negative bacteria. The bactericidal action of cefprozil results from inhibition of cell-wall synthesis. Cefprozil has been shown to be active against most strains of the following microorganisms both in vitro and in clinical infections as described in the INDICATIONS AND USAGE section.
Aerobic Gram-Positive Microorganisms
Staphylococcus aureus
(including β-lactamase-producing strains)
NOTE: Cefprozil is inactive against methicillin-resistant staphylococci.
Streptococcus pneumoniae
Streptococcus pyogenes
Aerobic Gram-Negative Microorganisms
Haemophilus influenzae
(including β-lactamase-producing strains)
Moraxella (Branhamella) catarrhalis
(including β-lactamase-producing strains)
The following in vitro data are available; however, their clinical significance is unknown. Cefprozil exhibits in vitro minimum inhibitory concentrations (MICs) of 8 mcg/mL or less against most (≥90%) strains of the following microorganisms; however, the safety and effectiveness of cefprozil in treating clinical infections due to these microorganisms have not been established in adequate and well-controlled clinical trials.
Aerobic Gram-Positive Microorganisms
Enterococcus durans
Enterococcus faecalis
Listeria monocytogenes
Staphylococcus epidermidis
Staphylococcus saprophyticus
Staphylococcus warneri
Streptococcus agalactiae
Streptococci (Groups C, D, F, and G)
viridans group Streptococci
NOTE: Cefprozil is inactive against Enterococcus faecium
Aerobic Gram-Negative Microorganisms
Citrobacter diversus
Escherichia coli
Klebsiella pneumoniae
Neisseria gonorrhoeae
(including β-lactamase-producing strains)
Proteus mirabilis
Salmonella spp.
Shigella spp.
Vibrio spp.
NOTE: Cefprozil is inactive against most strains of Acinetobacter, Enterobacter, Morganella morganii, Proteus vulgaris, Providencia, Pseudomonas, andSerratia.
Anaerobic Microorganisms
Prevotella (Bacteroides) melaninogenicus
Clostridium difficile
Clostridium perfringens
Fusobacterium spp.
Peptostreptococcus spp.
Propionibacterium acnes
NOTE: Most strains of the Bacteroides fragilis group are resistant to cefprozil.
Susceptibility Tests
Dilution Techniques
Quantitative methods are used to determine antimicrobial minimal inhibitory concentrations (MICs). These MICs provide estimates of the susceptibility of bacteria to antimicrobial compounds. The MICs should be determined using a standardized procedure. Standardized procedures are based on a dilution method1,2 (broth or agar) or equivalent with standardized inoculum concentrations and standardized concentrations of cefprozil powder. The MIC values should be interpreted according to the following criteria:
A report of “Susceptible” indicates that the pathogen is likely to be inhibited if the antimicrobial compound in the blood reaches the concentrations usually achievable. A report of “Intermediate” indicates that the result should be considered equivocal, and, if the microorganism is not fully susceptible to alternative, clinically feasible drugs, the test should be repeated. This category implies possible clinical applicability in body sites where the drug is physiologically concentrated or in situations where high dosage of drug can be used. This category also provides a buffer zone which prevents small uncontrolled technical factors from causing major discrepancies in interpretation. A report of “Resistant” indicates that the pathogen is not likely to be inhibited if the antimicrobial compound in the blood reaches the concentrations usually achievable; other therapy should be selected.
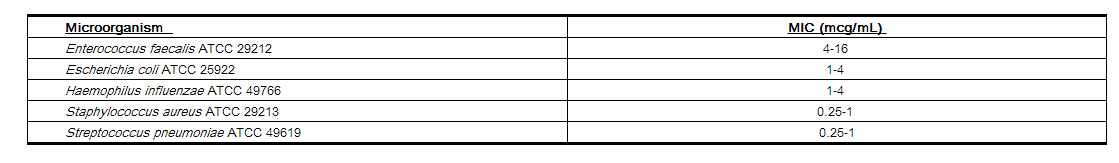
Standardized susceptibility test procedures require the use of laboratory control microorganisms to control the technical aspects of the laboratory procedures. Standard cefprozil powder should provide the following MIC values:
Diffusion Techniques
Quantitative methods that require measurement of zone diameters also provide reproducible estimates of the susceptibility of bacteria to antimicrobial compounds. One such standardized procedure3 requires the use of standardized inoculum concentrations. This procedure uses paper disks impregnated with 30 mcg cefprozil to test the susceptibility of microorganisms to cefprozil.
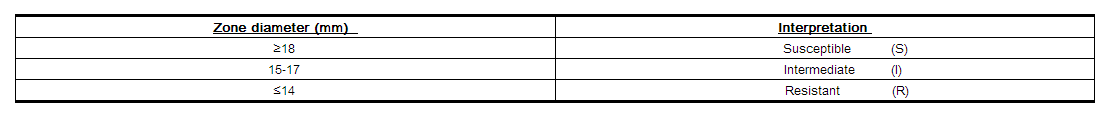
Reports from the laboratory providing results of the standard single-disk susceptibility test with a 30 mcg cefprozil disk should be interpreted according to the following criteria:
Interpretation should be as stated above for results using dilution techniques. Interpretation involves correlation of the diameter obtained in the disk test with the MIC for cefprozil.
As with standardized dilution techniques, diffusion methods require the use of laboratory control microorganisms that are used to control the technical aspects of the laboratory procedures. For the diffusion technique, the 30 mcg cefprozil disk should provide the following zone diameters in these laboratory test quality control strains.
References
http://www.accessdata.fda.gov/drugsatfda_docs/label/2007/050664s024,050665s024lbl.pdf