Topiramate dosage and administration
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Pratik Bahekar, MBBS [2]
For patient information, click here.
Dosage and Administration
Epilepsy
It is not necessary to monitor topiramate plasma concentrations to optimize topiramate tablets therapy.
On occasion, the addition of topiramate tablets to phenytoin may require an adjustment of the dose of phenytoin to achieve optimal clinical outcome. Addition or withdrawal of phenytoin and/or carbamazepine during adjunctive therapy with topiramate tablets may require adjustment of the dose of topiramate tablets.
Because of the bitter taste, tablets should not be broken.
Topiramate tablets can be taken without regard to meals.
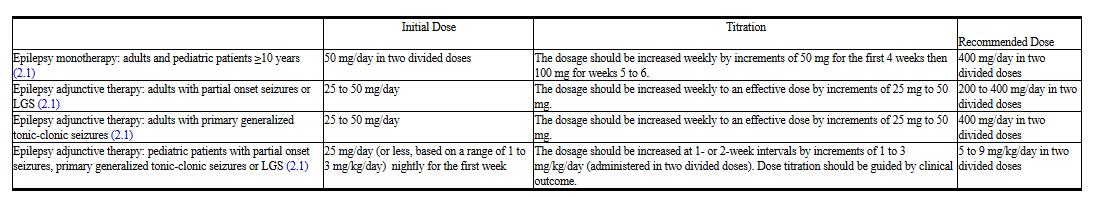
Monotherapy Use
Adults and Pediatric Patients 10 Years and Older
The recommended dose for topiramate monotherapy in adults and pediatric patients 10 years of age and older is 400 mg/day in two divided doses. Approximately 58% of patients randomized to 400 mg/day achieved this maximal dose in the monotherapy controlled trial; the mean dose achieved in the trial was 275 mg/day. The dose should be achieved by titration according to the following schedule (Table 1):
References
- ↑ "TOPIRAMATE (TOPIRAMATE ) TABLET, FILM COATED [AUROBINDO PHARMA LIMITED]". Retrieved 6 February 2014.
Template:Antimigraine preparations
 |