Rifampin description
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Chetan Lokhande, M.B.B.S [2]
Description
RIFADIN (rifampin capsules USP) for oral administration contain 150 mg or 300 mg rifampin per capsule. The 150 mg and 300 mg capsules also contain, as inactive ingredients: corn starch, D&C Red No. 28, FD&C Blue No. 1, FD&C Red No. 40, gelatin, magnesium stearate, and titanium dioxide.
RIFADIN IV (rifampin for injection USP) contains rifampin 600 mg, sodium formaldehyde sulfoxylate 10 mg, and sodium hydroxide to adjust pH.
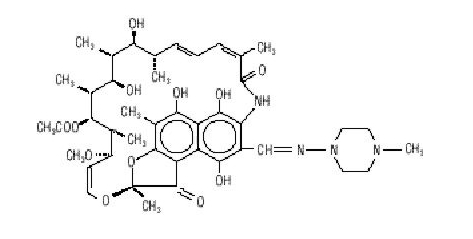
Rifampin is a semisynthetic antibiotic derivative of rifamycin SV. Rifampin is a red-brown crystalline powder very slightly soluble in water at neutral pH, freely soluble in chloroform, soluble in ethyl acetate and in methanol. Its molecular weight is 822.95 and its chemical formula is C43H58N4O12. The chemical name for rifampin is either:
3-[[(4-Methyl-1-piperazinyl)imino]methyl]rifamycin
or
5,6,9,17,19,21-hexahydroxy-23-methoxy-2,4,12,16,20,22– heptamethyl-8-[N-(4-methyl-1-piperazinyl)formimidoyl]-2,7-(epoxypentadeca[1,11,13]trienimino)naphtho[2,1-b]furan-1,11(2H)-dione 21-acetate.
Its structural formula is:[1]
 |
References
Adapted from the FDA Package Insert.