Ventricular fibrillation pathophysiology: Difference between revisions
(Created page with "__NOTOC__ {{Ventricular fibrillation}} {{CMG}} ==Overview== ==Pathophysiology== Ventricular fibrillation has been described as "chaotic asynchronous fractionated activity of...") |
|||
| Line 3: | Line 3: | ||
{{CMG}} | {{CMG}} | ||
==Overview== | ==Overview== | ||
Ventricular fibrillation is a cause of [[cardiac arrest]] and [[sudden cardiac death]]. The ventricular muscle twitches randomly rather than contracting in a coordinated fashion (from the apex of the heart to the outflow of the ventricles), and so the [[ventricle (heart)|ventricle]]s fail to pump blood into the [[artery|arteries]] and [[systemic circulation]]. Ventricular fibrillation is a sudden lethal arrhythmia responsible for many deaths in the Western world, and it is mostly caused by [[ischemic heart disease]]. While most episodes occur in diseased hearts, others can afflict normal hearts as well. | |||
Despite considerable research, the underlying nature of ventricular fibrillation is still not completely understood. | |||
==Pathophysiology== | ==Pathophysiology== | ||
Revision as of 13:32, 4 September 2012
|
Ventricular fibrillation Microchapters |
|
Differentiating Ventricular Fibrillation from other Diseases |
|---|
|
Diagnosis |
|
Treatment |
|
Case Studies |
|
Ventricular fibrillation pathophysiology On the Web |
|
American Roentgen Ray Society Images of Ventricular fibrillation pathophysiology |
|
Risk calculators and risk factors for Ventricular fibrillation pathophysiology |
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]
Overview
Ventricular fibrillation is a cause of cardiac arrest and sudden cardiac death. The ventricular muscle twitches randomly rather than contracting in a coordinated fashion (from the apex of the heart to the outflow of the ventricles), and so the ventricles fail to pump blood into the arteries and systemic circulation. Ventricular fibrillation is a sudden lethal arrhythmia responsible for many deaths in the Western world, and it is mostly caused by ischemic heart disease. While most episodes occur in diseased hearts, others can afflict normal hearts as well.
Despite considerable research, the underlying nature of ventricular fibrillation is still not completely understood.
Pathophysiology
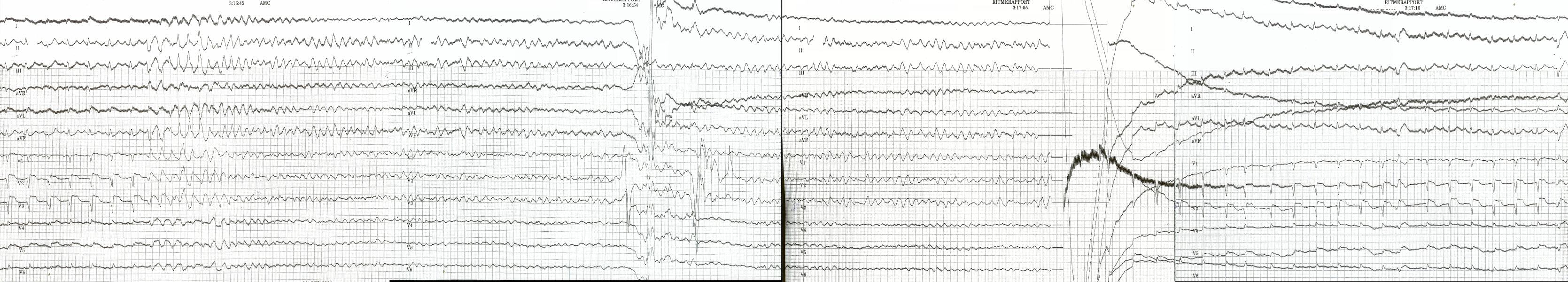
Ventricular fibrillation has been described as "chaotic asynchronous fractionated activity of the heart" (Moe et al. 1964). A more complete definition is that ventricular fibrillation is a "turbulent, disorganized electrical activity of the heart in such a way that the recorded electrocardiographic deflections continuously change in shape, magnitude and direction".[1]
Ventricular fibrillation most commonly occurs within diseased hearts, and, in the vast majority of cases, is a manifestation of underlying ischemic heart disease. Ventricular fibrillation is also seen in those with cardiomyopathy, myocarditis, and other heart pathologies. In addition, it is seen with electrolyte disturbances and overdoses of cardiotoxic drugs. It is also notable that ventricular fibrillation occurs where there is no discernible heart pathology or other evident cause, the so-called idiopathic ventricular fibrillation.
Idiopathic ventricular fibrillation occurs with a reputed incidence of approximately 1% of all cases of out-of-hospital arrest, as well as 3%-9% of the cases of ventricular fibrillation unrelated to myocardial infarction, and 14% of all ventricular fibrillation resuscitations in patients under the age of 40.[2] It follows then that, on the basis of the fact that ventricular fibrillation itself is common, idiopathic ventricular fibrillation accounts for an appreciable mortality. Recently-described syndromes such as the Brugada Syndrome may give clues to the underlying mechanism of ventricular arrhythmias. In the Brugada syndrome, changes may be found in the resting ECG with evidence of right bundle branch block (RBBB) and ST elevation in the chest leads V1-V3, with an underlying propensity to sudden cardiac death.[3]
The relevance of this is that theories of the underlying pathophysiology and electrophysiology must account for the occurrence of fibrillation in the apparent "healthy" heart. It is evident that there are mechanisms at work that we do not fully appreciate and understand. Investigators are exploring new techniques of detecting and understanding the underlying mechanisms of sudden cardiac death in these patients without pathological evidence of underlying heart disease.[4]
Familial conditions that predispose individuals to developing ventricular fibrillation and sudden cardiac death are often the result of gene mutations that affect cellular transmembrane ion channels. For example, in Brugada Syndrome, sodium channels are affected. In certain forms of long QT syndrome, the potassium inward rectifier channel is affected.
Triggered activity
Triggered activity can occur due to the presence of afterdepolarisations. These are depolarising oscillations in the membrane voltage induced by preceding action potentials. These can occur before or after full repolarisation of the fiber and as such are termed either early (EADs) or delayed afterdepolarisations (DADs). All afterdepolarisations may not reach threshold potential, but, if they do, they can trigger another afterdepolarisation, and thus self-perpetuate.
Characteristics of the ventricular fibrillation waveform
Ventricular fibrillation can be described in terms of its electrocardiographic waveform appearance. All waveforms can be described in terms of certain features, such as amplitude and frequency. Researchers have looked at the frequency of the ventricular fibrillation waveform to see if it helps to elucidate the underlying mechanism of the arrhythmia or holds any clinically useful information. More recently, Gray has suggested an underlying mechanism for the frequency of the waveform that has puzzled investigators as possibly being a manifestation of the Doppler effect of rotors of fibrillation.[5] Analysis of the fibrillation waveform is performed using a mathematical technique known as Fourier analysis.
Power spectrum
The distribution of frequency and power of a waveform can be expressed as a power spectrum in which the contribution of different waveform frequencies to the waveform under analysis is measured. This can be expressed as either the dominant or peak frequency, i.e., the frequency with the greatest power or the median frequency, which divides the spectrum in two halves.
Frequency analysis has many other uses in medicine and in cardiology, including analysis of heart rate variability and assessment of cardiac function, as well as in imaging and acoustics.[6][7]

References
- ↑ Robles de Medina EO, Bernard R, Coumel P; et al. (1978). "Definition of terms related to cardiac rhythm. WHO/ISFC Task Force". Eur J Cardiol. 8 (2): 127–44. PMID 699945.
- ↑ Viskin S, Belhassen B (1990). "Idiopathic ventricular fibrillation". Am. Heart J. 120 (3): 661–71. doi:10.1016/0002-8703(90)90025-S. PMID 2202193.
- ↑ Brugada P, Brugada J (1992). "Right bundle branch block, persistent ST segment elevation and sudden cardiac death: a distinct clinical and electrocardiographic syndrome. A multicenter report". J. Am. Coll. Cardiol. 20 (6): 1391–6. doi:10.1016/0735-1097(92)90253-J. PMID 1309182.
- ↑ Saumarez RC, Heald S, Gill J; et al. (1995). "Primary ventricular fibrillation is associated with increased paced right ventricular electrogram fractionation". Circulation. 92 (9): 2565–71. PMID 7586358.
- ↑ Jalife J, Gray RA, Morley GE, Davidenko JM (1998). "Self-organization and the dynamical nature of ventricular fibrillation". Chaos. 8 (1): 79–93. doi:10.1063/1.166289. PMID 12779712.
- ↑ Shusterman V, Beigel A, Shah SI; et al. (1999). "Changes in autonomic activity and ventricular repolarization". J Electrocardiol. 32. Suppl: 185–92. doi:10.1016/S0022-0736(99)90078-X. PMID 10688324.
- ↑ Kaplan SR, Bashein G, Sheehan FH; et al. (2000). "Three-dimensional echocardiographic assessment of annular shape changes in the normal and regurgitant mitral valve". Am. Heart J. 139 (3): 378–87. doi:10.1016/S0002-8703(00)90077-2. PMID 10689248.