Nimesulide
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [2]
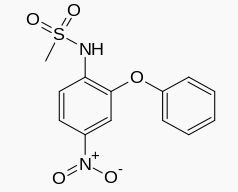 | |
| Clinical data | |
|---|---|
| AHFS/Drugs.com | International Drug Names |
| Routes of administration | Oral, rectal, topical |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Protein binding | >97.5% |
| Metabolism | Hepatic |
| Elimination half-life | 1.8–4.7h |
| Excretion | Renal (50%), fecal (29%) |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| E number | {{#property:P628}} |
| ECHA InfoCard | {{#property:P2566}}Lua error in Module:EditAtWikidata at line 36: attempt to index field 'wikibase' (a nil value). |
| Chemical and physical data | |
| Formula | C13H12N2O5S |
| Molar mass | 308.311 g/mol |
| 3D model (JSmol) | |
| |
| |
| | |
Overview
Nimesulide is a relatively COX-2 selective, non-steroidal anti-inflammatory drug (NSAID) with analgesic and antipyretic properties. Its approved indications are the treatment of acute pain, the symptomatic treatment of osteoarthritis and primary dysmenorrhoea in adolescents and adults above 12 years old. It has a multifactorial mode of action and is characterized by a fast onset of action.
History
It was launched in Italy for the first time as Aulin and Mesulid in 1985 and is available in more than 50 countries worldwide, including among others France, Portugal, Greece, Switzerland, Belgium, Russia Thailand and Brazil. Nimesulide has never been filed for Food and Drug Administration (FDA) evaluation in the United States, where it is not marketed.[1] Due to concerns about the risk of hepatotoxicity, nimesulide has been withdrawn from market in many countries.Template:Which
Advantages
- Nimesulide has similar gastrointestinal safety as compared to other NSAIDs.[citation needed]
- Its multifactorial mode of action gives it a unique and broad action on inflammatory processes.[citation needed]
- It may be taken through prescription in the treatment of Acute (short term) pain, and Primary dysmenorrhea (Period Pains). It should never be taken in Painful osteoarthritis (Swelling in the joints) .[2]
Availability
It is available in a variety of forms: tablets, powder for dissolution in water, suppositories, mouth dissolving tablets and topical gel.
A recent evaluation from the European Medicines Agency (EMA) concluded that the overall benefit/risk profile of nimesulide is favourable and in line with that of the other NSAIDs such as diclofenac, ibuprofen, and naproxen.
Trade names
Nimesulide is available through the world as original product with the following trademarks: Sulide, Nimalox, Mesulid (Novartis, Brazil, Boehringer Ingelheim, Greece), Coxtal (Sanofi, China, Czech, Bulgaria),Sintalgin (Abbott, Brazil), Eskaflam (GSK, Brazil, Mexico), Octaprin, Nimside (Teva,Pakistan), Nise (Dr. Reddy’s Laboratories, India, Russia, Venezuela, Vietnam, Ukraine), Nilsid (Egypt); Aulin, Ainex, Drexel, Donulide, Edrigyl, Enetra, Heugan, Mesulid, Minapon, NeRelid, Nexen, Nidolon, Nilden (Mexico); Nimed, Nimedex, Nimesil, Nimulid, Nimutab, Nimdase, Nimopen-MP (India), Nisulid, Nodard Plus, Nicip, Nimcap, Nic-P, Nic-Spas (India); Novolid, Relmex (Ecuador); Remisid (Ukraine); Scaflam, Scaflan, Sulidin (Turkey), Xilox (Hungary); Modact-IR (Pakistan);[3] Sulidene and Zolan for veterinary use. Many generic and copy-products also exist (Lusemin, Medicox, Nidol, Nimalox, Nimesil, Nimotas, Nimulid, Nizer, Sorini, Ventor, Vionim, Neolide, Willgo among others), new-aid (S.A.R.), Nims (Nepal).
Pharmacokinetics
Nimesulide is rapidly absorbed following oral administration.[4]
Nimesulide undergoes extensive biotransformation, mainly to 4-hydroxynimesulide (which also appears to be biologically active).[4]
Food, gender and advanced age have negligible effects on nimesulide pharmacokinetics.[4]
Moderate renal impairment does not necessitate dosage adjustment while patients with severe renal impairment or hepatic impairment are contraindicated.[5]
Nimesulide has a relatively rapid onset of action, with meaningful reductions in pain and inflammation observed within 15 minutes from drug intake.[6][7]
The therapeutic effects of Nimesulide are the result of its complete mode of action which targets a number of key mediators of the inflammatory process such as: COX-2 mediated prostaglandins, free radicals, proteolytic enzymes and histamine.[6] Clinical evidence is available to support a particularly good profile in terms of gastrointestinal tolerability.[8]
Nimesulide Events
Madras High Court revokes suspension on manufacture and sale of Nimesulide in India
On September 13, 2011 Madras High Court revoked a suspension on manufacture and sale of paediatric drugs nimesulide and phenylpropanolamine (PPA).[9]
EMEA re-confirms efficacy of Nimesulide
On 23 June 2011, the European Medicines Agency concluded the review of systemic nimesulide-containing medicine. They concluded that the benefits of systemic nimesulide continue to outweigh their risks in the treatment of patients with acute pain and primary dysmenorrhea.
Central Drugs Standard Control Organization of India Restricts use of Nimesulide under 12 years of age.
Several reports have been made of adverse drug reactions in India.[10][11][12][13] On Feb 12, 2011, Express India that the Union Ministry of Health and Family Welfare had finally decided to suspend the pediatric use of the analgesic, Nimesulide suspension.[14] From 10 March 2011 onwards Nimesulide formulations not indicated for human use in children below 12 years of age.[15]
EMA confirms the positive benefit/risk ratio
On September 21, 2007 the EMA released a press release on their review on the liver-related safety of nimesulide. The EMA has concluded that the benefits of these medicines outweigh their risks, but that there is a need to limit the duration of use to ensure that the risk of patients developing liver problems is kept to a minimum. Therefore the EMA has limited the use of systemic formulations (tablets, solutions, suppositories) of nimesulide to 15 days.[16]
Irish Medicines Board (IMB) suspends Nimesulide containing drugs (15 May 2007)
The Irish Medicines Board (IMB) has decided to suspend Nimesulide from the Irish market and refer it to the EU Committee for Human Medicinal Products (CHMP) for a review of its benefit/risk profile. The decision is due to the reporting of six (6) cases of potentially related liver failures to the IMB by the National Liver Transplant Unit, St Vincent Hospital. These cases occurred in the period from 1999 to 2006.[17]
Delhi High Court verdict
In response to a petition against Nimesulide filed in May 2004, the Delhi High Court passed a verdict that Nimesulide will continue to be marketed in India since the concerns over its side effect profile are unsubstantiated. The High court sought the help of leading pediatricians and medical agencies from India to arrive at the conclusion that Nimesulide is a safe NSAID with a side effect profile that is comparable to the other NSAIDs.
Bribes allegedly paid in Italy to spare few drugs from official scrutiny
In May 2008, Italy's leading daily paper Corriere della Sera and other media outlets[citation needed] reported that a top-ranking official at Italy's medicines agency AIFA had been filmed by police while accepting bribes from employees of pharmaceutical companies.[18][19] The money was allegedly being paid to ensure that certain drugs would be spared scrutiny from the drugs watchdog. The investigation had started in 2005 following suspicions that some AIFA drug tests had been faked. Eight arrests were made. Nimesulide can be bought carrying a prescription from a physician that is kept as a receipt at the chemist shop, nominally allowing strong control over selling.
The original manufacturer of Nimesulide is Helsinn Healthcare SA, Switzerland, which acquired the rights for the drug in 1976. After the patent protection had terminated, a number of other companies have started production and marketing of Nimesulide.
European Medicines Agency reports increased risk of liver toxicity
The Committee noted that the studies looking into the effectiveness of nimesulide in acute pain relief have shown that it is as effective as other NSAID pain killers such as diclofenac, ibuprofen and naproxen.
In terms of safety, the Committee noted that nimesulide has the same risk of causing stomach and gut problems as other NSAIDs. To limit the risk of side effects affecting the liver, several restrictions have already been introduced in the past, including restriction to second line treatment, the use of lowest effective doses for the shortest possible duration, and a maximum duration of treatment for acute pain.
The CHMP concluded that nimesulide was associated with an increased risk of liver toxicity compared with other anti-inflammatory treatments. The CHMP is now recommending, as a further restriction, that systemic nimesulide should no longer be used for treating painful osteoarthritis. The Committee considered that the use of systemic nimesulide for the treatment of painful osteoarthritis, which is a chronic condition, will increase the risk of the medicines being used for long-term treatment with a consequent increase in the risk of liver injury.
Side effects
The use of nimesulide in children under the age of 12 is contraindicated. Continuous use of nimesulide (more than 15 days) can cause the following side effects:
- Diarrhea
- Vomiting
- Skin rash
- Pruritis
- Dizziness
- Bitterness in mouth
Women should use the drug with caution during lactation and it is contraindicated during pregnancy.[20]
References
- ↑ Traversa G, Bianchi C, Da Cas R, Abraha I, Menniti-Ippolito F, Venegoni M (July 2003). "Cohort study of hepatotoxicity associated with nimesulide and other non-steroidal anti-inflammatory drugs". BMJ. 327 (7405): 18–22. doi:10.1136/bmj.327.7405.18. PMC 164233. PMID 12842950.
- ↑ "Current status: European Commission final decision". Retrieved 12 November 2014.
- ↑ [1]
- ↑ 4.0 4.1 4.2 Bernareggi A (October 1998). "Clinical pharmacokinetics of nimesulide". Clin Pharmacokinet. 35 (4): 247–74. doi:10.2165/00003088-199835040-00001. PMID 9812177.
- ↑ Microsoft Word - opnh.P.Nimesulide .EMEA-CPMP-3086-03-en-Final.doc
- ↑ 6.0 6.1 Rainsford KD (June 2006). "Nimesulide – a multifactorial approach to inflammation and pain: scientific and clinical consensus". Curr Med Res Opin. 22 (6): 1161–70. doi:10.1185/030079906X104849. PMID 16846549.
- ↑ Bianchi M, Broggini M (2003). "A randomised, double-blind, clinical trial comparing the efficacy of nimesulide, celecoxib and rofecoxib in osteoarthritis of the knee". Drugs. 63 (Suppl 1): 37–46. doi:10.2165/00003495-200363001-00006. PMID 14506910.
- ↑ Laporte JR, Ibáñez L, Vidal X, Vendrell L, Leone R (2004). "Upper gastrointestinal bleeding associated with the use of NSAIDs: newer versus older agents". Drug Saf. 27 (6): 411–20. doi:10.2165/00002018-200427060-00005. PMID 15144234.
- ↑ Madras High Court Revokes Suspension on Manufacture and Sale PPA
- ↑ Safety of nimesulide. CD ROM, Appropriate Use of Antipyretics / Analgesics in Children, Health Informatics, New Delhi, 2004.
- ↑ Rahman SZ, Khan RA (2004). "Is nimesulide safe in a cardiovascular-Compromised patient?". Indian J Pharmacol. 36: 252–3.
- ↑ Khan RA, Rahman SZ (2004). "A Case Report on Nimesulide and its Relation with Angina". J Pharmacovigilance Drug Safety. 1: 19–21.
- ↑ Khan RA, Rahman SZ (2004). "Nimesulide Induced Coronary Artery Insufficiency – A Case Report". J Pharmacovigilance Drug Safety. 1: 11–3.
- ↑ Nimesulide for kids to be suspended finally
- ↑ CDSCO website-wide gazette notification GSR 82(E) dated 10.03.2011.
- ↑ EMA press release on nimesulide September 2007
- ↑ IMB Announces Immediate Suspension of the Marketing of Medicines Containing Nimesulide
- ↑ «Mazzette per evitare i controlli sull'Aulin». Mario Pappagallo, Corriere della Sera, 23 May 2008[dead link]
- ↑ Italian medicines agency officials arrested in corruption probe. Manufacturing Chemist
- ↑ http://www.pharmaceutical-drug-manufacturers.com/pharmaceutical-drugs/nimesulide.html
- Pages with script errors
- CS1 maint: Multiple names: authors list
- All articles with dead external links
- Articles with dead external links from June 2014
- Articles with invalid date parameter in template
- Template:drugs.com link with non-standard subpage
- Drugs with non-standard legal status
- Articles with changed DrugBank identifier
- Articles with changed EBI identifier
- E number from Wikidata
- ECHA InfoCard ID from Wikidata
- Chemical articles with unknown parameter in Infobox drug
- Drugboxes which contain changes to verified fields
- All articles with unsourced statements
- Articles with unsourced statements from December 2012
- Articles with unsourced statements from June 2014
- Non-steroidal anti-inflammatory drugs
- Antipyretics
- Analgesics
- Phenol ethers