Carotene
Overview

The term carotene is used for several related substances having the formula C40H56. Carotene is an orange photosynthetic pigment important for photosynthesis. It is responsible for the orange colour of the carrot and many other fruits and vegetables. It contributes to photosynthesis by transmitting the light energy it absorbs to chlorophyll.
Chemically, carotene is a terpene, synthesized biochemically from eight isoprene units. It comes in two primary forms designated by characters from the Greek alphabet: alpha-carotene (α-carotene) and beta-carotene (β-carotene). Gamma, delta and epsilon (γ, δ and ε-carotene) also exist. Beta-carotene is composed of two retinyl groups, and is broken down in the mucosa of the small intestine by beta-carotene dioxygenase to retinol, a form of vitamin A. Carotene can be stored in the liver and converted to vitamin A as needed, thus making it a provitamin.
Dietary sources

The following foods are particularly rich in carotenes:
- Sweet potatoes[1]
- carrots[1]
- kale[1]
- spinach[1]
- cantaloupe melon[2]
- turnip greens[1]
- winter squash[1]
- collard greens[1]
- cilantro[1]
- fresh thyme[1]
- Romaine lettuce[1]
- goji berries
- Ivy gourd
- Cassava
- mango
Absorption from these foods is enhanced if eaten with fats, as carotenes are fat soluble, and if the food is cooked for a few minutes until the plant cell wall splits and the colour is released into any liquid.
The two forms
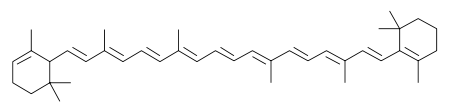

The two primary isomers of carotene, α-carotene and β-carotene, differ in the position of double bonds in the cyclic group at the end.
β-Carotene is the more common form and can be found in yellow, orange, and green leafy fruits and vegetables. These can be carrots, spinach, lettuce, tomatoes, sweet potatoes, broccoli, cantaloupe, oranges, and winter squash. As a rule of thumb, the greater the intensity of the colour of the fruit or vegetable, the more β-carotene it contains.
β-carotene is present in yellow orange bell peppers (called yellow capsicum in Australia) but is not the principal colour in red bell peppers, chillis and other varieties and species in the genus Capsicum. A wide range of carotenoids and other colourful compounds abound in the plant kingdom. We can attribute benefits to the plant in expending resources to produce these compounds from their role in attracting insects for pollination and alluring animals for seed distribution, to protecting vital cell functions against the destructive effects of ultraviolet light.
β-Carotene is an anti-oxidant and as such can be useful for curbing the excess of damaging free radicals in the body. However, the usefulness of β-carotene as a dietary supplement (i.e. taken as a pill) in cancer prevention is still subject to debate.[3] β-Carotene is fat-soluble.
Beta-carotene and cancer
It has been shown in trials that the use of synthetically-produced beta carotene (that is, beta carotene in supplement form such as the pills typically sold in stores) increases the rate of lung cancer and prostate cancer, and increases mortality in smokers. These results have been observed in beta carotene supplements and not in foods with naturally occurring beta carotene.[4]
An article on the American Cancer Society says that The Cancer Research Campaign has called for warning labels on beta carotene supplements to caution smokers that such supplements may increase the risk of lung cancer.[5]
The New England Journal of Medicine published an article[6] in 1994 about a trial which examined the relationship between daily supplementation of beta carotene and vitamin E (alpha-tocopherol) and the incidence of lung cancer. The study was done using supplements and researchers were aware of the relationship between carotenoid-rich fruits and vegetables and lower lung cancer rates. The research concluded that no reduction in lung cancer was found in the participants using these supplements (beta-carotene), and furthermore, these supplements may, in fact, have harmful effects.
The Journal of the National Cancer Institute published an article in 1996[7] about a trial that was conducted to determine if vitamin A (in the form of retinyl palmitate) and beta carotene had any beneficial effects to prevent cancer. The results indicate an increased risk of lung cancer for the participants who consumed the beta-carotene supplement.
A review of all randomized controlled trials in the scientific literature by the Cochrane Collaboration published in JAMA in 2007 found that beta carotene increased mortality by 5% (Relative Risk 1.05, 95% confidence interval 1.01-1.08).[8]
Carotenemia
Carotene is often labeled as good for you. However, your body converts this yellow pigment to Vitamin A, and too much Vitamin A can be harmful to your body. Carotenemia or hypercarotenemia is excess carotene, but unlike excess vitamin A, carotene is non-toxic. Although hypercarotenemia is not particularly dangerous, it can lead to a yellowing of the skin (carotenodermia). It is most commonly associated with consumption of an abundance of carrots, but it also can be a medical sign of more dangerous conditions. A randomised trial into the use of β-carotene and vitamin A for prevention of lung cancer had to be stopped early due to the apparent increase in the incidence of lung cancer in those with lung irritation from smoking or asbestos exposure.[3]
Production
Most of the world's synthetic supply of carotene comes from a manufacturing complex located in Freeport, Texas and owned by DSM. In Spain Vitatene produces natural beta carotene form Blakeslea trispora. In Australia, organic beta-carotene is produced by Aquacarotene Limited from dried marine algae Dunaliella salina grown in harvesting ponds situated in Karratha, Western Australia.
Carotene is also found in corn, and in the milk of Guernsey dairy cows. Carotene causes the milk of the Guernsey cow to turn yellow.
Total synthesis
There are currently two commonly used methods of total synthesis of β-carotene. The first was developed by the Badische Anilin- & Soda-Fabrik (BASF) and is based on the Wittig reaction. The second is a Grignard reaction, elaborated by Hoffman-La Roche from the original synthesis of Inhoffen et al. They are both symmetrical; the BASF synthesis is C20 + C20 , and the Hoffman-La Roche synthesis is C19 + C2 + C19.
Nomenclature
Carotenes are carotenoids containing no oxygen. Carotenoids containing some oxygen are known as xanthophylls.
The two ends of the β-carotene molecule are structurally identical, and are called β-rings. Specifically, the group of nine carbon atoms at each end form a β-ring.
The α-carotene molecule has a β-ring at one end; the other end is called an ε-ring. There is no such thing as an "α-ring".
These and similar names for the ends of the carotenoid molecules form the basis of a systematic naming scheme, according to which:
- α-carotene is β,ε-carotene;
- β-carotene is β,β-carotene;
- γ-carotene (with one β ring and one uncyclized end that is labelled psi) is β,ψ-carotene;
- δ-carotene (with one ε ring and one uncyclized end) is ε,ψ-carotene;
- ε-carotene is ε,ε-carotene
6 μg of dietary β-carotene supplies the equivalent of 1 μg of retinol, or 1 RE (Retinol Equivalent). This is equivalent to 3⅓ IU of vitamin A.
Use in Medications
Pill Images
{{#ask: Page Name::Carotene |?Pill Name |?Drug Name |?Pill Ingred |?Pill Imprint |?Pill Dosage |?Pill Color |?Pill Shape |?Pill Size (mm) |?Pill Scoring |?NDC |?Drug Author |format=template |template=DrugPageImages |mainlabel=- |sort=Pill Name }} {{#subobject:
|Page Name=Carotene |Pill Name=PRENATAL_PLUS_IRON_NDC_09045339.jpg |Drug Name=PRENATAL PLUS IRON |Pill Ingred=VITAMIN A ACETATE[VITAMIN A];BETA-CAROTENE[.BETA-CAROTENE];ASCORBIC ACID[ASCORBIC ACID];CHOLECALCIFEROL[CHOLECALCIFEROL];ALPHA-TOCOPHEROL ACETATE;DL-[Alpha-TOCOPHEROL;DL-];THIAMINE MONONITRATE[Thiamine ION];RIBOFLAVIN[RIBOFLAVIN];NIACINAMIDE[NIACINAMIDE];PYRIDOXINE HYDROCHLORIDE[PYRIDOXINE];FOLIC ACID[FOLIC ACID];CYANOCOBALAMIN[CYANOCOBALAMIN];CALCIUM CARBONATE[CALCIUM CATION];IRON PENTACARBONYL[IRON PENTACARBONYL];ZINC OXIDE[ZINC OXIDE];CUPRIC OXIDE[CUPRIC OXIDE]|+sep=; |Pill Imprint=CIS28;G13 |Pill Dosage=920 [iU] |Pill Color=Yellow|+sep=; |Pill Shape=Oval |Pill Size (mm)=18 |Pill Scoring=1 |Pill Image= |Drug Author=Major Pharmaceuticals |NDC=09045339
}}
{{#subobject:
|Page Name=Carotene |Pill Name=PRENATAL_PLUS_NDC_651620668.jpg |Drug Name=PRENATAL PLUS |Pill Ingred=VITAMIN A ACETATE[VITAMIN A];BETA-CAROTENE[.BETA-CAROTENE];ASCORBIC ACID[ASCORBIC ACID];CHOLECALCIFEROL[CHOLECALCIFEROL];ALPHA-TOCOPHEROL ACETATE;DL-[Alpha-TOCOPHEROL;DL-];THIAMINE MONONITRATE[Thiamine ION];RIBOFLAVIN[RIBOFLAVIN];NIACINAMIDE[NIACINAMIDE];PYRIDOXINE HYDROCHLORIDE[PYRIDOXINE];FOLIC ACID[FOLIC ACID];CYANOCOBALAMIN[CYANOCOBALAMIN];CALCIUM CARBONATE[CALCIUM CATION];FERROUS FUMARATE[FERROUS CATION];ZINC OXIDE[ZINC OXIDE];CUPRIC OXIDE[CUPRIC CATION]|+sep=; |Pill Imprint=G12 |Pill Dosage=920 [iU] |Pill Color=Yellow|+sep=; |Pill Shape=Oval |Pill Size (mm)=19 |Pill Scoring=1 |Pill Image= |Drug Author=Amneal Pharmaceuticals, LLC |NDC=651620668
}}
External links
- Beta-carotene website by Martha Evens, School of Chemistry, University of Bristol
- Berkeley Wellness Guide to Dietary Supplements
- Beta-carotene on University of Maryland
- Carotene at the US National Library of Medicine Medical Subject Headings (MeSH)
References
- ↑ 1.00 1.01 1.02 1.03 1.04 1.05 1.06 1.07 1.08 1.09 What can foods rich in beta-carotene do for you?
- ↑ [1]
- ↑ 3.0 3.1 Omenn GS, Goodman GE, Thornquist MD; et al. (1996). "Effects of a combination of beta carotene and vitamin A on lung cancer and cardiovascular disease". N. Engl. J. Med. 334 (18): 1150–5. PMID 8602180.
- ↑ Beta Carotene, Mayo Clinic
- ↑ "British Cancer Organization Calls for Warning Labels on Beta-Carotene". 2000-07-31. Retrieved 2007-03-15.
- ↑ The Alpha-Tocopherol, Beta Carotene Cancer Prevention Study Group (1994). "The effect of vitamin E and beta carotene on the incidence of lung cancer and other cancers in male smokers". N. Engl. J. Med. 330 (15): 1029–35. PMID 8127329.
- ↑ Omenn GS, Goodman GE, Thornquist MD; et al. (1996). "Risk factors for lung cancer and for intervention effects in CARET, the Beta-Carotene and Retinol Efficacy Trial". J. Natl. Cancer Inst. 88 (21): 1550–9. PMID 8901853.
- ↑ Bjelakovic G, Nikolova D, Gluud LL, Simonetti RG, Gluud C (2007). "Mortality in randomized trials of antioxidant supplements for primary and secondary prevention: systematic review and meta-analysis". JAMA. 297 (8): 842–57. doi:10.1001/jama.297.8.842. PMID 17327526.
ar:كاروتين bg:Каротин cs:Karoteny de:Carotine eo:Karoteno id:Karoten it:Carotene lt:Karotinas mk:Каротин nl:Caroteen nn:Karoten sv:Karoten Template:Jb1