Becaplermin
Editor-In-Chief: C. Michael Gibson, M.S., M.D. [1]; Associate Editor(s)-in-Chief: Vignesh Ponnusamy, M.B.B.S. [2]
Disclaimer
WikiDoc MAKES NO GUARANTEE OF VALIDITY. WikiDoc is not a professional health care provider, nor is it a suitable replacement for a licensed healthcare provider. WikiDoc is intended to be an educational tool, not a tool for any form of healthcare delivery. The educational content on WikiDoc drug pages is based upon the FDA package insert, National Library of Medicine content and practice guidelines / consensus statements. WikiDoc does not promote the administration of any medication or device that is not consistent with its labeling. Please read our full disclaimer here.
Black Box Warning
|
WARNING
See full prescribing information for complete Boxed Warning.
INCREASED RATE OF MORTALITY SECONDARY TO MALIGNANCY:
|
Overview
Becaplermin is a human platelet-derived growth factor that is FDA approved for the treatment of lower extremity diabetic neuropathic ulcers that extend into the subcutaneous tissue or beyond and have an adequate blood supply. There is a Black Box Warning for this drug as shown here. Common adverse reactions include erythematous rashes.
Adult Indications and Dosage
FDA-Labeled Indications and Dosage (Adult)
Diabetic Neuropathic Ulcer
- For topical use; not for oral, ophthalmic or intravaginal use.
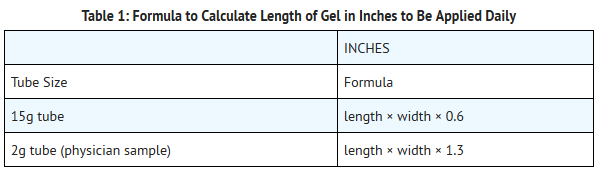
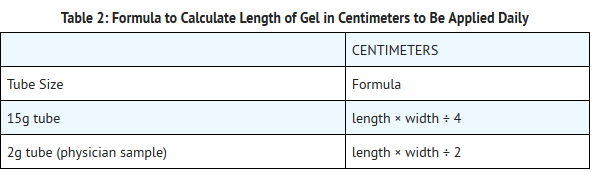
- The amount of REGRANEX Gel to be applied will vary depending upon the size of the ulcer area. To calculate the length of gel to apply to the ulcer, measure the greatest length of the ulcer by the greatest width of the ulcer in either inches or centimeters. To calculate the length of gel in inches, use the formula shown below in Table 1, and to calculate the length of gel in centimeters, use the formula shown below in Table 2.
- Using the calculation, each square inch of ulcer surface will require approximately 2/3 inch length of gel squeezed from a 15g tube, or approximately 1 1/3 inch length of the gel from a 2g tube (physician sample). For example, if the ulcer measures 1 inch by 2 inches, then a 1 1/4 inch length of gel should be used for 15g tubes (1 × 2 × 0.6 = 1 1/4) and 2 3/4 inch gel length should be used for a 2g tube (1 × 2 × 1.3 = 2 3/4).
- Using the calculations for ulcer size in centimeters, each square centimeter of ulcer surface will require approximately a 0.25 centimeter length of gel squeezed from a 15g tube, or approximately a 0.5 centimeter length of gel from a 2g tube. For example, if the ulcer measures 4 cm by 2 cm, then a 2 centimeter length of gel should be used for a 15g tube [(4 × 2) ÷ 4 = 2] and a 4 centimeter length of gel should be used for a 2g tube [(4 × 2) ÷ 2 = 4].
Off-Label Use and Dosage (Adult)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Becaplermin in adult patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Becaplermin in adult patients.
Pediatric Indications and Dosage
FDA-Labeled Indications and Dosage (Pediatric)
- Safety and effectiveness of REGRANEX Gel in pediatric patients below the age of 16 years have not been established.
Off-Label Use and Dosage (Pediatric)
Guideline-Supported Use
There is limited information regarding Off-Label Guideline-Supported Use of Becaplermin in pediatric patients.
Non–Guideline-Supported Use
There is limited information regarding Off-Label Non–Guideline-Supported Use of Becaplermin in pediatric patients.
Contraindications
- REGRANEX Gel is contraindicated in patients with known neoplasm(s) at the site(s) of application.
Warnings
|
WARNING
See full prescribing information for complete Boxed Warning.
INCREASED RATE OF MORTALITY SECONDARY TO MALIGNANCY:
|
Precautions
- Cancer and Cancer Mortality
- REGRANEX Gel contains becaplermin, a recombinant human platelet-derived growth factor, which promotes cellular proliferation and angiogenesis. The benefits and risks of becaplermin treatment should be carefully evaluated before prescribing. Becaplermin should be used with caution in patients with a known malignancy.
- Malignancies distant from the site of application have occurred in becaplermin users in both a clinical study and postmarketing use, and an increased rate of death from systemic malignancies was seen in patients who have received 3 or more tubes of REGRANEX Gel.
- In a follow-up study, 491 (75%) of 651 subjects from two randomized, controlled trials of becaplermin gel 0.01% were followed for a median of approximately 20 months to identify malignancies diagnosed after the end of the trials. Eight of 291 subjects (3%) from the becaplermin group and two of 200 subjects (1%) from the vehicle/standard of care group were diagnosed with cancers during the follow-up period, a relative risk of 2.7 (95% confidence interval 0.6–12.8). The types of cancers varied and all were remote from the treatment site.
- In a retrospective study of a medical claims database, cancer rates and overall cancer mortality were compared between 1,622 patients who used REGRANEX Gel and 2,809 matched comparators. Estimates of the incidence rates reported below may be under-reported due to limited follow-up for each individual.
- The incidence rate for all cancers was 10.2 per 1,000 person years for patients treated with REGRANEX Gel and 9.1 per 1,000 person years for the comparators. Adjusted for several possible confounders, the rate ratio was 1.2 (95% confidence interval 0.7–1.9). Types of cancers varied and were remote from the site of treatment.
- The incidence rate for mortality from all cancers was 1.6 per 1,000 person years for those who received REGRANEX Gel and 0.9 per 1,000 person years for the comparators. The adjusted rate ratio was 1.8 (95% confidence interval 0.7–4.9).
- The incidence rate for mortality from all cancers among patients who received 3 or more tubes of REGRANEX Gel was 3.9 per 1,000 person years and 0.9 per 1,000 person years in the comparators. The adjusted rate ratio for cancer mortality among those who received 3 or more tubes relative to those who received none was 5.2 (95% confidence interval 1.6–17.6).
- Application Site Reactions
- If application site reactions occur, the possibility of sensitization or irritation caused by parabens or m-cresol should be considered. Consider interruption or discontinuation and further evaluation (e.g. patch testing) as dictated by clinical circumstances.
Adverse Reactions
Clinical Trials Experience
- Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in practice.
- In a follow-up study from two randomized, controlled trials, an increased rate of cancer remote from the becaplermin treatment site was observed in subjects treated with REGRANEX Gel.
- In clinical trials, erythematous rashes occurred in 2% of patients treated with REGRANEX Gel (and good ulcer care) or placebo (and good ulcer care), and none in patients receiving good ulcer care alone. Patients treated with REGRANEX Gel did not develop neutralizing antibodies against becaplermin.
Postmarketing Experience
- An increased rate of mortality secondary to malignancy was observed in patients treated with 3 or more tubes of REGRANEX Gel in a postmarketing retrospective cohort study.
- Burning sensation at the site of application and erythema have been reported during post-approval use of REGRANEX Gel. Because post approval adverse reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to the drug.
Drug Interactions
- It is not known if REGRANEX Gel interacts with other topical medications applied to the ulcer site. The use of REGRANEX Gel with other topical drugs has not been studied.
Use in Specific Populations
Pregnancy
- Pregnancy Category C
- There are no adequate and well-controlled studies in pregnant women treated with REGRANEX Gel. REGRANEX Gel should be used during pregnancy only if the potential benefit justifies the potential risk to the fetus. Animal reproduction studies have not been conducted with REGRANEX Gel.
- Australian Drug Evaluation Committee (ADEC) Pregnancy Category
There is no Australian Drug Evaluation Committee (ADEC) guidance on usage of Becaplermin in women who are pregnant.
Labor and Delivery
There is no FDA guidance on use of Becaplermin during labor and delivery.
Nursing Mothers
- It is not known whether becaplermin is excreted in human milk. Because many drugs are secreted in human milk, caution should be exercised when REGRANEX Gel is administered to nursing women.
Pediatric Use
- Safety and effectiveness of REGRANEX Gel in pediatric patients below the age of 16 years have not been established.
Geriatic Use
- Among patients receiving any dose of REGRANEX Gel in clinical studies of diabetic lower extremity ulcers, 150 patients were 65 years of age and older. No overall differences in safety or effectiveness were observed between patients < 65 years of age and patients ≥ 65 years of age. The number of patients aged 75 and older were insufficient (n=34) to determine whether they respond differently from younger patients.
Gender
There is no FDA guidance on the use of Becaplermin with respect to specific gender populations.
Race
There is no FDA guidance on the use of Becaplermin with respect to specific racial populations.
Renal Impairment
There is no FDA guidance on the use of Becaplermin in patients with renal impairment.
Hepatic Impairment
There is no FDA guidance on the use of Becaplermin in patients with hepatic impairment.
Females of Reproductive Potential and Males
There is no FDA guidance on the use of Becaplermin in women of reproductive potentials and males.
Immunocompromised Patients
There is no FDA guidance one the use of Becaplermin in patients who are immunocompromised.
Administration and Monitoring
Administration
- Topical
Monitoring
There is limited information regarding Monitoring of Becaplermin in the drug label.
IV Compatibility
There is limited information regarding IV Compatibility of Becaplermin in the drug label.
Overdosage
Chronic Overdose
There is limited information regarding Chronic Overdose of Becaplermin in the drug label.
Pharmacology
Becaplermin
| |
| Systematic (IUPAC) name | |
| ? | |
| Identifiers | |
| CAS number | |
| ATC code | A01 , D03AX06 (WHO) |
| PubChem | ? |
| DrugBank | |
| Chemical data | |
| Formula | ? |
| Mol. mass | ? |
| Pharmacokinetic data | |
| Bioavailability | ? |
| Metabolism | ? |
| Half life | ? |
| Excretion | ? |
| Therapeutic considerations | |
| Pregnancy cat. |
C |
| Legal status |
Rx only |
| Routes | Topical |
Mechanism of Action
- REGRANEX Gel has biological activity similar to that of endogenous platelet-derived growth factor, which includes promoting the chemotactic recruitment and proliferation of cells involved in wound repair and enhancing the formation of granulation tissue.
Structure
- REGRANEX Gel contains becaplermin, a recombinant human platelet-derived growth factor for topical administration. Becaplermin is produced by recombinant DNA technology by insertion of the gene for the B chain of platelet-derived growth factor (PDGF) into the yeast, Saccharomyces cerevisiae. Becaplermin has a molecular weight of approximately 25 KD and is a homodimer composed of two identical polypeptide chains that are bound together by disulfide bonds. REGRANEX Gel is a non-sterile, low bioburden, preserved, sodium carboxymethylcellulose-based (CMC) topical gel, containing the active ingredient becaplermin and the following inactive ingredients: carboxymethylcellulose sodium, glacial acetic acid, l-lysine hydrochloride, m-cresol, methylparaben, propylparaben, sodium acetate trihydrate, sodium chloride, and water for injection. Each gram of REGRANEX Gel contains 100 mcg of becaplermin.
Pharmacodynamics
There is limited information regarding Pharmacodynamics of Becaplermin in the drug label.
Pharmacokinetics
- Ten patients with Stage III or IV (as defined in the International Association of Enterostomal Therapy (IAET) guide to chronic wound staging,1, 2 lower extremity diabetic ulcers received topical applications of becaplermin gel 0.01% at a dose range of 0.32–2.95 µg/kg (7µg/cm2) daily for 14 days. Six patients had non-quantifiable PDGF levels at baseline and throughout the study, two patients had PDGF levels at baseline which did not increase substantially, and two patients had PDGF levels that increased sporadically above their baseline values during the 14 day study period.
Nonclinical Toxicology
Carcinogenesis, Mutagenesis, Impairment of Fertility
- Becaplermin was not genotoxic in a battery of in vitro assays (including those for bacterial and mammalian cell point mutation, chromosomal aberration, and DNA damage/repair). Becaplermin was also not mutagenic in an in vivo assay for the induction of micronuclei in mouse bone marrow cells.
- Carcinogenesis and reproductive toxicity studies have not been conducted with REGRANEX Gel.
Animal Toxicology and/or Pharmacology
- In nonclinical studies, rats injected at the metatarsals with 3 or 10 mcg/site (approximately 60 or 200 mcg/kg) of becaplermin every other day for 13 days displayed histological changes indicative of accelerated bone remodeling consisting of periosteal hyperplasia and subperiosteal bone resorption and exostosis. The soft tissue adjacent to the injection site had fibroplasia with accompanying mononuclear cell infiltration reflective of the ability of PDGF to stimulate connective tissue growth.
Clinical Studies
- The effects of REGRANEX Gel on the incidence of and time to complete healing in lower extremity diabetic ulcers were assessed in four randomized controlled studies. Of 922 patients studied, 478 received either REGRANEX Gel 0.003% or 0.01%. All study participants had lower extremity diabetic neuropathic ulcers that extended into the subcutaneous tissue or beyond (Stages III and IV of the IAET guide to chronic wound staging). Ninety-three percent of the patients enrolled in these four trials had foot ulcers. The remaining 7% of the patients had ankle or leg ulcers. The diabetic ulcers were of at least 8 weeks duration and had an adequate blood supply (defined as TcpO2 > 30 mm Hg). In the four trials, ninety-five percent of the ulcers measured in area up to 10 cm2, and the median ulcer size at baseline ranged from 1.4 cm2 to 3.5 cm2. All treatment groups received a program of good ulcer care consisting of initial complete sharp debridement, a non-weight-bearing regimen, systemic treatment for wound-related infection if present, moist saline dressings changed twice a day, and additional debridement as necessary. REGRANEX Gel 0.003% or 0.01% or placebo gel was applied once a day and covered with a saline moistened dressing. After approximately 12 hours, the gel was gently rinsed off and a saline moistened dressing was then applied for the remainder of the day. Patients were treated until complete healing, or for a period of up to 20 weeks. Patients were considered a treatment failure if their ulcer did not show an approximately 30% reduction in initial ulcer area after eight to ten weeks of REGRANEX Gel therapy.
- The primary endpoint, incidence of complete ulcer closure within 20 weeks, for all treatment arms is shown in Figure 1. In each study, REGRANEX Gel in conjunction with good ulcer care was compared to placebo gel plus good ulcer care or good ulcer care alone.
- In Study 1, a multicenter, double-blind, placebo controlled trial of 118 patients, the incidence of complete ulcer closure for REGRANEX Gel 0.003% (n=61) was 48% versus 25% for placebo gel (n=57; p=0.02, logistic regression analysis).
- In Study 2, a multicenter, double-blind, placebo controlled trial of 382 patients, the incidence of complete ulcer closure for REGRANEX Gel 0.01% (n=123) was 50% versus 36% for REGRANEX Gel 0.003% (n=132) and 35% for placebo gel (n=127). Only REGRANEX Gel 0.01% was significantly different from placebo gel (p=0.01, logistic regression analysis).
- The primary goal of Study 3, a multicenter controlled trial of 172 patients, was to assess the safety of vehicle gel (placebo; n=70) compared to good ulcer care alone (n=68). The study included a small (n=34) REGRANEX Gel 0.01% arm. Incidences of complete ulcer closure were 44% for REGRANEX Gel, 36% for placebo gel and 22% for good ulcer care alone.
- In Study 4, a multicenter, evaluator-blind, controlled trial of 250 patients, the incidences of complete ulcer closure in the REGRANEX Gel 0.01% arm (n=128) (36%) and good ulcer care alone (n=122) (32%) were not statistically different.
- In general, where REGRANEX Gel was associated with higher incidences of complete ulcer closure, differences in the incidence first became apparent after approximately 10 weeks and increased with continued treatment (Table 3).
- In a 3-month follow-up period where no standardized regimen of preventative care was utilized, the incidence of ulcer recurrence was approximately 30% in all treatment groups, demonstrating that the durability of ulcer closure was comparable in all treatment groups.
- In a randomized, double-blind study of REGRANEX Gel (100 mcg/g once daily for 16 weeks) in patients with Stage III or IV pressure ulcers, the incidence of complete ulcer closure was 15% (28/189) in the becaplermin group and 12% (22/190) in the vehicle control group. This difference was not statistically significant.
- In two small, randomized, double-blinded studies of REGRANEX Gel (100 mcg/g once daily for 16 weeks) in patients with venous stasis ulcers, the combined incidence of complete ulcer closure was 46% (30/65) in the becaplermin group and 39% (26/67) in the vehicle control group. This difference was not statistically significant.
How Supplied
- REGRANEX Gel is available in multi-use tubes in the following size:
- 15 g tube NDC 0064-0810-15
- REGRANEX Gel is for external use only.
- Store refrigerated at 2° – 8° C (36° – 46°F). Do not freeze. Do not use the gel after the expiration date shown at the bottom of the tube.
Storage
There is limited information regarding Becaplermin Storage in the drug label.
Images
Drug Images
{{#ask: Page Name::Becaplermin |?Pill Name |?Drug Name |?Pill Ingred |?Pill Imprint |?Pill Dosage |?Pill Color |?Pill Shape |?Pill Size (mm) |?Pill Scoring |?NDC |?Drug Author |format=template |template=DrugPageImages |mainlabel=- |sort=Pill Name }}
Package and Label Display Panel
{{#ask: Label Page::Becaplermin |?Label Name |format=template |template=DrugLabelImages |mainlabel=- |sort=Label Page }}
Patient Counseling Information
- Counsel patients to review and discuss any questions or concerns with their healthcare provider before starting REGRANEX and at regular intervals while receiving REGRANEX.
- Patients should be advised that:
- they should read the medication guide;
- hands should be washed thoroughly before applying REGRANEX Gel;
- the tip of the tube should not come into contact with the ulcer or any other surface; the tube should be recapped tightly after each use;
- a cotton swab, tongue depressor, or other application aid should be used to apply REGRANEX Gel;
- REGRANEX Gel should only be applied once a day in a carefully measured quantity. The measured quantity of gel should be spread evenly over the ulcerated area to yield a thin continuous layer of approximately 1/16 of an inch thickness. The measured length of the gel to be squeezed from the tube should be adjusted according to the size of the ulcer. The amount of REGRANEX Gel to be applied daily should be recalculated at weekly or biweekly intervals by the physician or wound care giver.
- Step-by-step instructions for application of REGRANEX Gel are as follows:
- Squeeze the calculated length of gel onto a clean, firm, nonabsorbable surface, e.g., wax paper.
- With a clean cotton swab, tongue depressor, or similar application aid, spread the measured REGRANEX Gel over the ulcer surface to obtain an even layer.
- Cover with a saline moistened gauze dressing.
- after approximately 12 hours, the ulcer should be gently rinsed with saline or water to remove residual gel and covered with a saline-moistened gauze dressing (without REGRANEX Gel);
- it is important to use REGRANEX Gel together with a good ulcer care program, including a strict non-weight-bearing program;
- excess application of REGRANEX Gel has not been shown to be beneficial;
- REGRANEX Gel should be stored in the refrigerator. Do not freeze REGRANEX Gel;
- REGRANEX Gel should not be used after the expiration date on the bottom, crimped end of the tube.
Precautions with Alcohol
- Alcohol-Becaplermin interaction has not been established. Talk to your doctor about the effects of taking alcohol with this medication.
Brand Names
- REGRANEX®[1]
Look-Alike Drug Names
- Regranex® — Granulex®[2]
Drug Shortage Status
Price
References
The contents of this FDA label are provided by the National Library of Medicine.
- ↑ "REGRANEX becaplermin gel".
- ↑ "http://www.ismp.org". External link in
|title=(help)
{{#subobject:
|Page Name=Becaplermin
|Pill Name=No image.jpg
|Drug Name=
|Pill Ingred=|+sep=;
|Pill Imprint=
|Pill Dosage={{{dosageValue}}} {{{dosageUnit}}}
|Pill Color=|+sep=;
|Pill Shape=
|Pill Size (mm)=
|Pill Scoring=
|Pill Image=
|Drug Author=
|NDC=
}}
{{#subobject:
|Label Page=Becaplermin |Label Name=Becaplermin05.png
}}
{{#subobject:
|Label Page=Becaplermin |Label Name=Becaplermin06.png
}}